Ground Rules
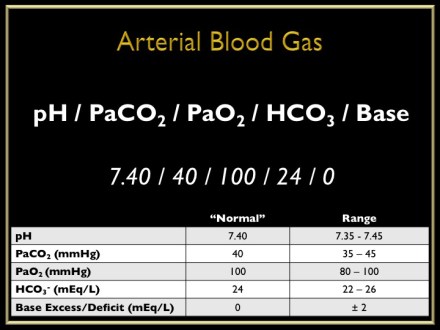
- Accurate assessment of a patient’s acid/base status requires a measurement of arterial pH and PCO2
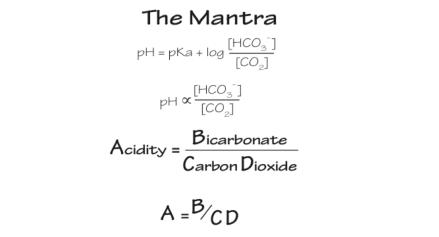
- bedside analyzers directly measure these and then use the Henderson-Hasselbach equation to calculate the HCO3-
- You can use venous blood, but must make adjustments:
- pH is 0.03-0.04 lower than arterial
- PCO2 is 7-8 mmHg higher than arterial
- HCO3- is 2 mEq/L higher than arterial
- A primary disturbance is usually accompanied by a compensatory response, but does not fully correct the pH
- bedside analyzers directly measure these and then use the Henderson-Hasselbach equation to calculate the HCO3-
There are four main ways pH can change:
- Metabolic Alkalosis
- increase in HCO3- and increase in pH
- Metabolic Acidosis
- Decrease in HCO3- and decrease in pH
- Respiratory Alkalosis
- Decrease in PCO2 and increase in pH
- Respiratory Acidosis
- Increase in PCO2 and decrease in pH
Remember, that the lungs can compensate considerable FASTER than the kidneys.
- Lungs can excrete 12 moles of acid per day as CO2
- Kidneys can excrete 0.1 moles of acid per day as ammonia, but can secrete HCO3- to buffer
Respiratory Acidosis
- Decrease in minute ventilation (TV x RR)
- Causes
- Normal Lungs
- CNS depression (drugs, head trauma)
- Neuromuscular impairment (GBS, MG)
- Thoracic restriction
- Abnormal Lungs
- Obstruction
- Alveoli dysfunction (ARDS)
- Perfusion defect (cardiac arrest, PTE)
- Normal Lungs
- Compensation
- Acute
- Increase of 10 mmHg of PaCO2 will increase serum HCO3- by 1 mEq/L
- Chronic
- Increase of 10 mmHg of PaCO2 will increase serum HCO3- by 3 mE1/L
- Acute
Respiratory Alkalosis
- Increase in minute ventilation (TV x RR)
- Causes
- Cardiac
- Hypoxemia
- Anemia
- Medications
- Pregnancy
- Iatrogenic
- Obstruction
- Neurologic
- Stress
- Compensation
- Acute
- Decrease of 10 mmHg of PaCO2 will decrease serum HCO3- by 2 mEq/L
- Chronic
- Decrease of 10 mmHg of PaCO2 will decrease serum HCO3- by 4 mEq/L
- Acute
If the condition is 100% acute respiratory, then the pH will change 0.08 for every 10 mmHg change in PaCO2
Metabolic Acidosis
- Decreased pH due to decreased HCO3-
- First step after determining that a patient has a metabolic acidosis is to calculate the anion gap
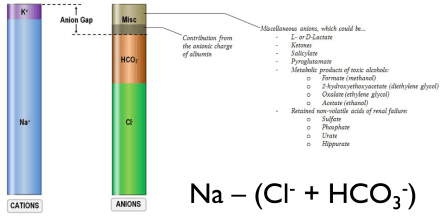
- High Anion Gap Metabolic Acidosis (HAGMA)
- Causes
- Carbon monoxide, cyanide
- Aminoglycosides
- Theophyline, toluene
- Methanol
- Uremia
- Diabetic ketoacidosis
- Propylene glycol
- Inborn errors of metabolism
- Lactic acidosis
- Ethylene glycol, ethanol
- Salicylates
- Causes
- Calculating a Delta Gap
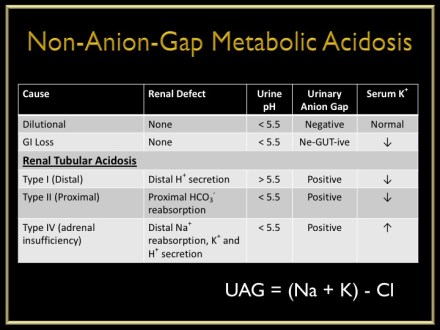
- Normal Anion Gap Metabolic Acidosis (NAGMA)
- Causes
- Ureteric diversion
- Small bowel fistulae
- Excessive saline
- Diarrhea
- Carbonic anhydrase inhibitors
- Renal tubular acidosis
- Adrenal insufficiency
- Pancreatic fistulae
- Causes
- Respiratory Compensation
- Expected PaCO2 = 8 + (1.5 x HCO3-) ± 2
Metabolic Alkalosis
- Increased pH due to increased HCO3-
- Causes
- Contraction
- Licorice
- Endocrine
- Vomiting
- Excessive NG suction
- Ringer’s solution
- Post-hypercapnia
- Diuretics
- Respiratory Compensation
- Expected PaCO2 = 20 + (0.7 x HCO3-) ± 5