Renal
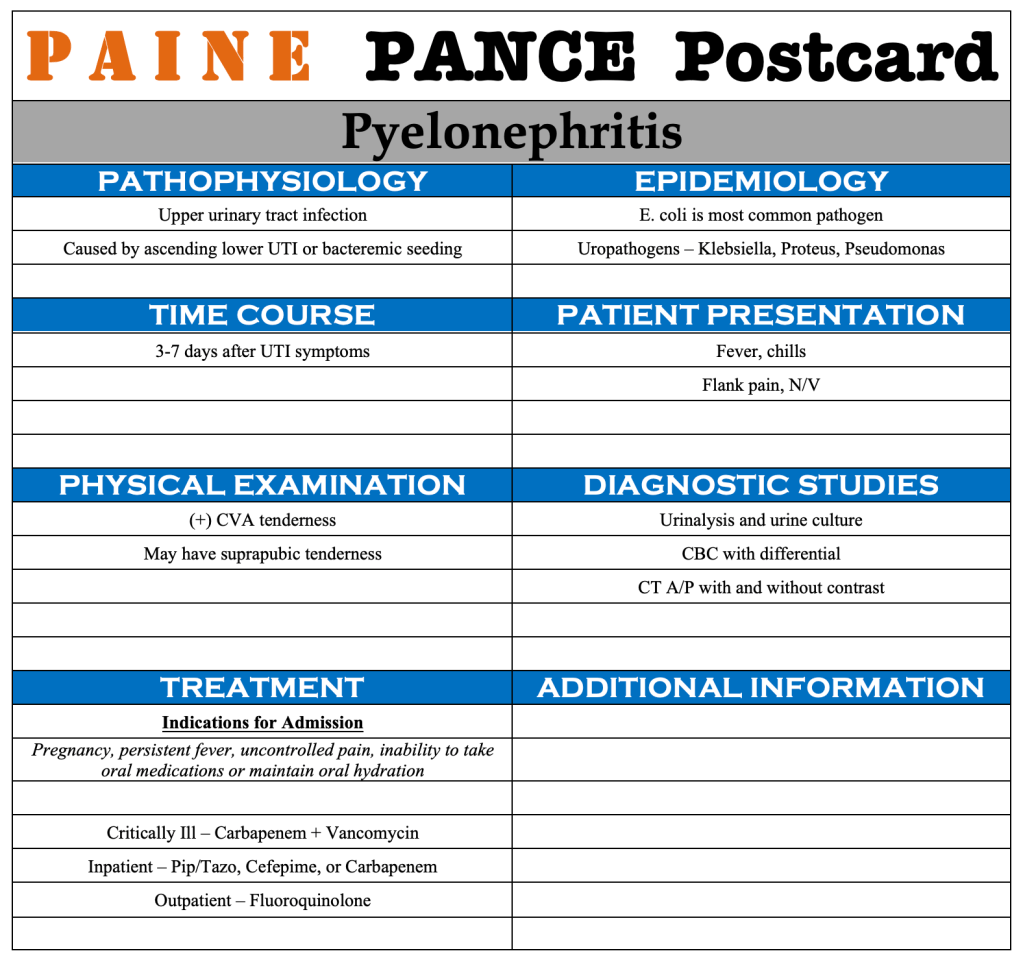
PAINE PANCE Postcard – Pyelonephritis

Diabetes Insipidus
***LISTEN TO THE PODCAST HERE***

Urinary Incontinence
***LISTEN TO THE PODCAST HERE***

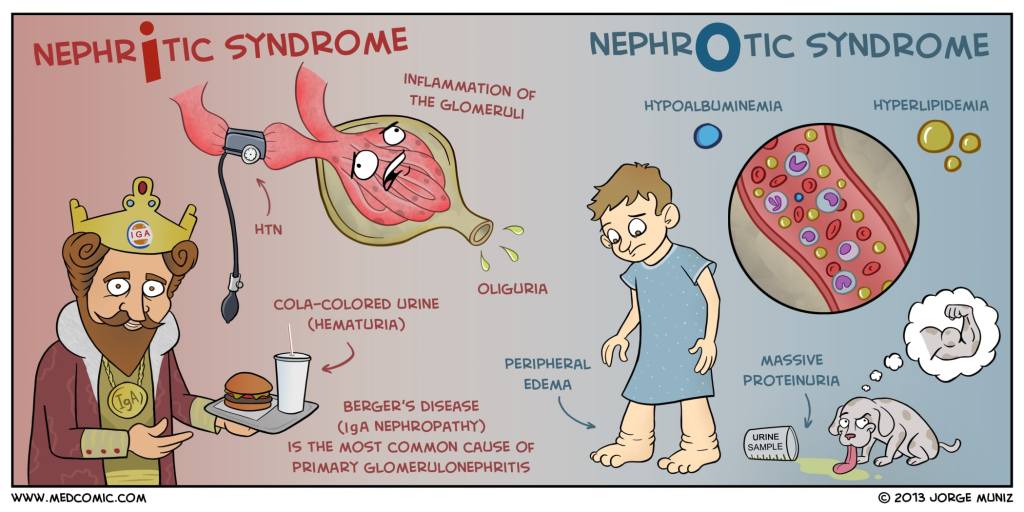
Nephrotic Syndrome
***LISTEN TO THE PODCAST HERE***
#64 – KDIGO Guidelines
***LISTEN TO THE PODCAST HERE***
What are KDIGO and KDOQI???
- The 2 Organizations
- Kidney Disease Outcomes Quality Initiative (KDOQI)
- US Based
- Developed in 1997 by National Kidney Foundation
- Kidney Disease: Improving Global Outcomes (KDIGO)
- Global organization developing and implementing evidence based clinical practice guidelines in kidney diseases
- Developed in 2003 by NKF
- Essentially individual entities, but both comment various aspects of kidney diseases
- Kidney Disease Outcomes Quality Initiative (KDOQI)
- 2012 Guidelines
- Published by KDIGO and commented by KDOQI
- 5 chapters
Chapter 1: Definition and Classification of CKD
- Definition
- Abnormalities in kidney structure or function, present for > 3 months, with implications on health





- Staging
- Based on causes, GFR category, and albuminuria category




- Predicting Prognosis of CKD

- Evaluation of GFR
- Recommend using serum creatinine and GFR estimating equation for initial assessment
- Recommend only using cystatin C in adult patients with decreased GFR but without markers of kidney damage if diagnosis of CKD is required

- Evaluation of Albuminuria
- Initial testing for proteinuria should be an early morning urine sample(in descending order of preference):
- Urine albumin-to-creatinine ratio (ACR)
- Urine protein-to-creatinine ratio (PCR)
- Reagent strip urinalysis for total protein with automated reading
- Reagent strip urinalysis for total protein with manual reading
- Microalbuminuria should no longer be used by laboratories
- If ACR > 30mg/g, then proceed to confirm with a random untimed urine sample
- Initial testing for proteinuria should be an early morning urine sample(in descending order of preference):
Chapter 2: Definition, Identification, and Predication of CKD Progression
- Assess albuminuria at least annually

- CKD progression is based on the one of the following:
- Decline in GFR category
- Drop in eGFR by ≥ 25% of baseline
- Sustained decline in eGFR by > 5mL/min/year
- Identify known risk factors associated with CKD progression
- Cause of CKD
- Age
- Gender
- Hypertension
- Hyperglycemia
- Dyslipidemia
- Smoking
- Obesity
- History of CVD
- Ongoing exposure to nephrotoxic agents
Chapter 3: Management of Progression and Complication of CKD
- Hypertension
- BP ≤ 140/90 if urine albumin excretion < 30mg/d
- BP ≤ 130/80 if urine albumin excretion > 30mg/d
- Recommend ACEI or ARB
- Protein Intake
- Recommend protein intake 0.8g/kg/d
- Glycemic Control
- Recommend HbA1C AROUND 7.0%
- ***newer ACE guidelines recommend < 6.5% with SGLT2i**

- Recommend < 2g/day
- Lifestyle
- Recommend 30 min/day five times per week, smoking cessation, and healthy weight (BMI 20-25)
- Lifestyle
- Complications Associated with CKD
- Anemia
- Diagnosed at < 13g/dL in men and < 12 g/dL in women
- Screening in patients with CKD:
- Stage G1-2 – when clinically indicated
- Stage 3a-3b – at least annually
- Stage 4-5 – at least twice per year
- Metabolic Bone Disease
- Obtained baseline calcium, phosphate, PTH, and ALP at least once in patients with GFR < 45 mL/min
- Not recommended to screen with bone mineral density testing
- Not recommended to supplement vitamin D of bisphophonates with deficiency or strong clinical rationale
- Acidosis
- Supplement oral bicarbonate in patients with serum bicarbonate < 22 mmol/L
- Anemia
Chapter 4: Other Complications of CKD
- CVD
- All CKD patients are at increased risk for CVD
- Recommend same testing and treating as non-CKD patients
- Use caution when interpreting NT-proBNP and troponins
- PVD
- Recommend regular podiatric assessment
- Medication Management
- Recommend using GFR for dosing adjustments
- Example – Metformin
- Stage G1-3a – continue
- Stage G3b – monitored
- Stage G4-5 – discontinued
- Example – Metformin
- Recommend using GFR for dosing adjustments

- Imaging studies and radiocontrast
- Avoid if possible, but do not hold if needed
- Following KDIGO Clinical Practice Guidelines for AKI
- Avoid high osmolar agents
- Use lowest contrast dose possible
- Stop nephrotoxic agents before and after
- Maintain adequate hydration
- Measure GFR 48-96 hours after
Chapter 5: Referral to Specialist and Models of Care



Cottage Physician (1893)

References
- KDIGO. Clinical Practice Guideline for the Evaluation and Management of CKD. 2012.
- Inker LA, Astor BC, Fox CH, et al. KDOQI US commentary on the 2012 KDIGO clinical practice guideline for the evaluation and management of CKD. Am J Kidney Dis. 2014; 63(5):713-35. [pubmed]
- Stevens PE, Levin A. Evaluation and management of chronic kidney disease: synopsis of the kidney disease: improving global outcomes 2012 clinical practice guideline. Ann Intern Med. 2013; 158(11):825-30. [pubmed]
- Andrassy KM. Comments on ‘KDIGO 2012 Clinical Practice Guideline for the Evaluation and Management of Chronic Kidney Disease’. Kidney Int. 2013; 84(3):622-3. [pubmed]
PAINE #PANCE Pearl – Surgery
Question
Surgery is a major physiologic stress and often is accompanied by biochemical derangements that effect homeostasis in the post-operative period. Describe the most common clinical scenarios that can cause each of the four main acid-base imbalances in a post-surgical patient.
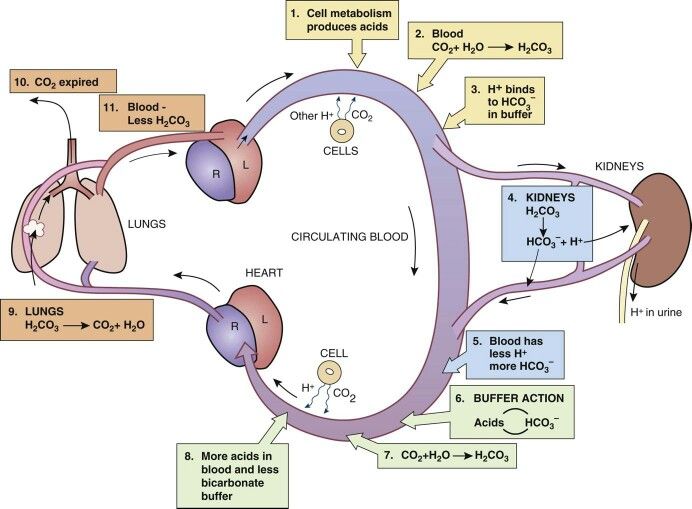
Answer
- Metabolic Acidosis
- Lactic acidosis (HAGMA)
- Under-resuscitation
- Blood loss
- Hyperchloremia (NAGMA)
- High chloride load from NaCl
- Lactic acidosis (HAGMA)
- Metabolic Alkalosis
- Volume contraction and bicarbonate reabsorption
- GI loss from NG tube suction or emesis
- Respiratory Acidosis
- Opioid medications causing depressed respiratory drive
- Respiratory Alkalosis
- Splinting from pain

PAINE #PANCE Pearl – Renal
Question
42yo woman, with a history systemic lupus erythematosus, presents to your clinic with a 1-month history of progressive leg swelling and polyuria. She is complaint with her medications and states that she hasn’t changed anything in her medical care. Physical examination reveals 2+ pitting edema to the knees in the lower extremities. BMP, UA, and urine microscopy are below.
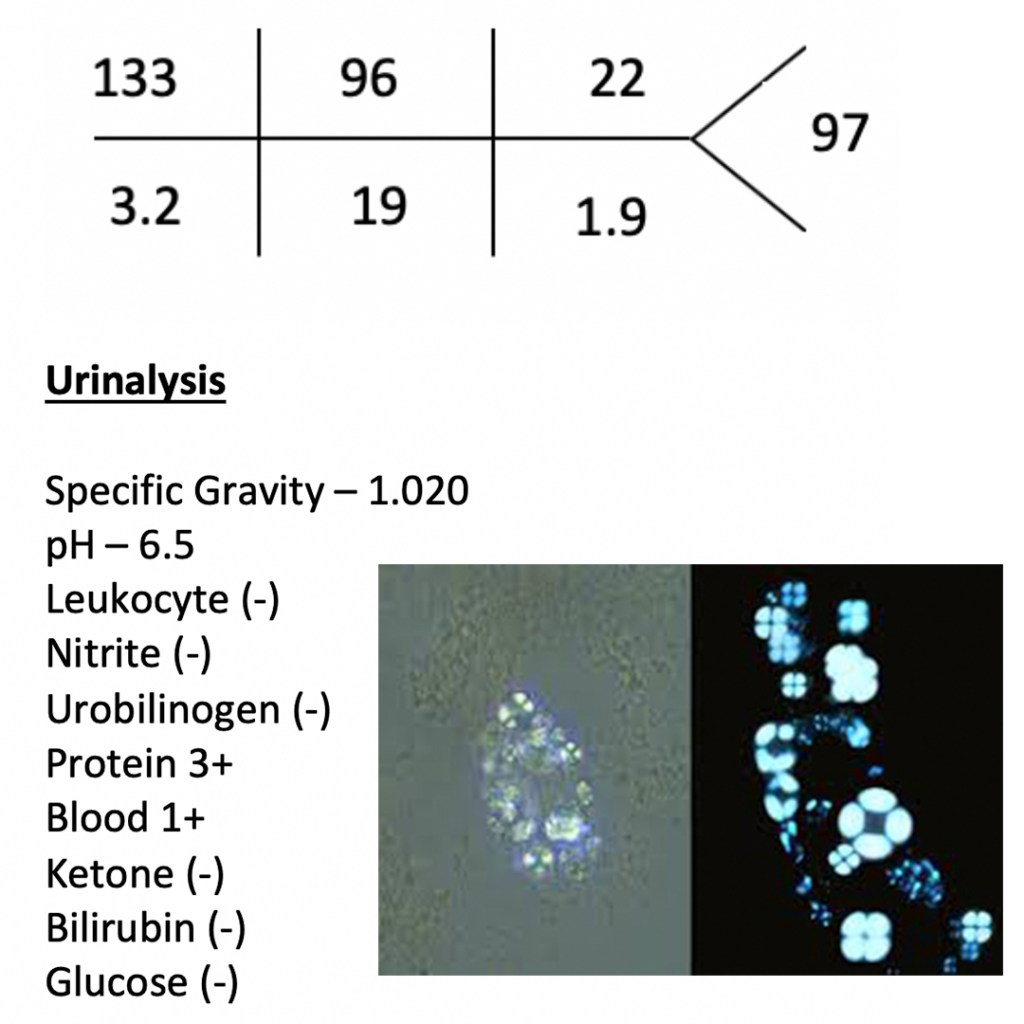
- What is the next step in diagnosing this patient and what would you expect to find?
Answer
This patient found to heavy proteinuria on a urinalysis and oval fat bodies on urine microscopy, which would point to nephrotic syndrome as a diagnosis.
The next step in the diagnostic management of this patient would be to perform a 24-hour urine collection for urine protein. Normal urine protein excretion is < 150mg/day, but nephrotic range proteinuria is diagnostic at > 3.5g/day. Alternatively, a random urine protein-to-creatinine ratio of > 3.5 can be used, but is less reliable than a 24-hour collection.
Once a nephrotic syndrome diagnosis is made by urine studies, it should be followed up with a renal biopsy to determine the cause.

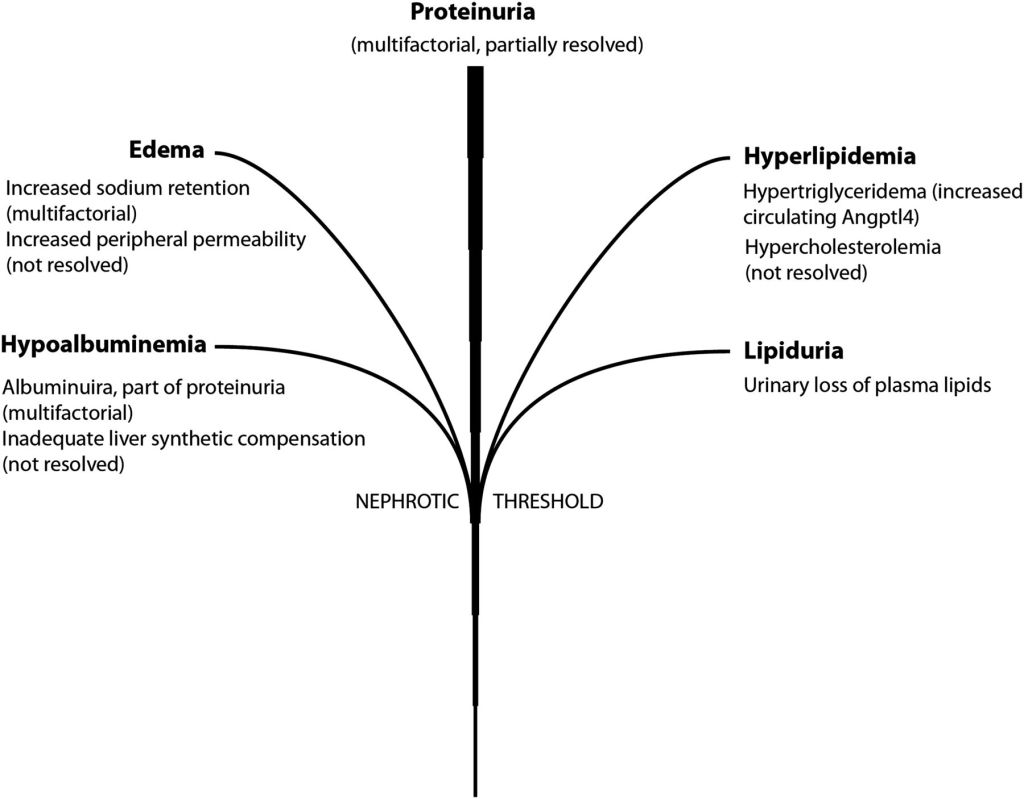
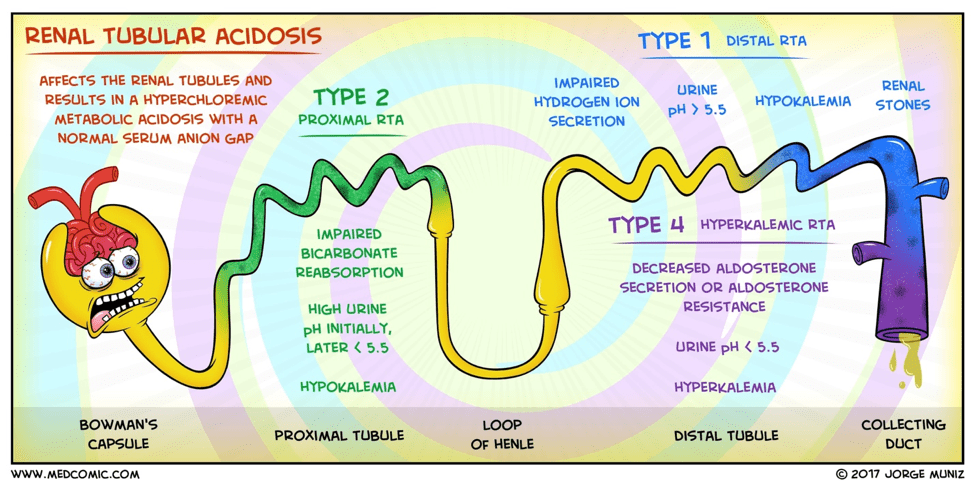
#51 – Renal Tubular Acidosis
***LISTEN TO THE PODCAST HERE***
Definition
- Group of disorders that cause a
metabolic acidosis due to defects in the renal tubules
- Net retention of HCl
- Net loss of NaHCO3
Pathophysiology
The kidney regulates acid-base balance two main ways:
- Reabsorption of filtered HCO3
- >80% of the bicarbonate filtered by the glomerulus is reabsorbed in the proximal renal tubules via Na-H exchange


- Acid excretion
- Collecting ducts of the nephron excrete hydrogen ions buffered by NH3 and PO3 (so the pH of the urine doesn’t destroy the nephron)
- Extra production of NH3 is stimulated by intracellular acidosis.
- Collecting ducts of the nephron excrete hydrogen ions buffered by NH3 and PO3 (so the pH of the urine doesn’t destroy the nephron)


- 3 step process
- Reabsorption of sodium to create a negative gradient in the tubular lumen
- Excretion of hydrogen by H-K-ATPase and reabsorption of potassium
- Prevention of hydrogen ions from diffusing back out of the tubular lumen
Initial Presentation
- Patients diagnosed with an RTA must first be diagnosed with a metabolic acidosis
- Decreased pH with decreased HCO3
- After this is determined, the anion gap must be calculated and found to be normal
- AG = Na – (Cl + HCO3) = 8-12
Differential for NAGMA
- Ureteric diversion
- Small bowel fistulae
- Excessive saline
- Diarrhea
- Carbonic anhydrase inhibitors
- Renal tubular acidosis
- Adrenal insufficiency
- Pancreatic fistulae
Type I (distal) RTA
- Cause
- Defect in the distal hydrogen ion excretion
- Pathophysiology
- Failure of the H-ATPase proton pump
(most common cause)
- Inability to acidify urine < 5.5
- Hypokalemia
- Increased hydrogen ion permeability of the luminal membrane
- Failure of the H-ATPase proton pump
(most common cause)

Type II (proximal) RTA
- Cause
- Defect in proximal bicarbonate reabsorption
- Pathophysiology
- Damage to the proximal tubule that leads to progressive bicarbonate wasting in the urine

Type IV (hypoaldosteronism)
- Cause
- Reductions in aldosterone secretion and responsiveness
- Pathophysiology
- Decreased rate of proton secretion rather than an intrinsic defect in the tubule’s capacity to generate normal pH gradient
- Hyperkalemia causes reduced urine
NH4, which in turns leads to more acidic urine
- Hydrogen ions have nothing to bind to
Diagnostic Work-Up
- RTAs should be considered in any
patient with a normal anion gap metabolic acidosis
- Need ABG and BMP
- Once this determination is made:
- Urine pH
- > 5.5 in type I (distal)
- < 5.5 in type II (proximal) and type IV
- Urine ammonium
- Elevated in type II (proximal)
- Decreased in type I (distal) and type IV
- Most labs can’t measure urine
ammonium directly:
- Urine Anion Gap (urine Na+K+Cl)
- (+) UAG = > 20
- Type I (distal) and type IV
- (-) UAG = < – 20
- Type II (proximal)
- (+) UAG = > 20
- Urine Anion Gap (urine Na+K+Cl)
- Serum potassium
- Elevated in type IV
- Decreased in type I and II
- Urine pH

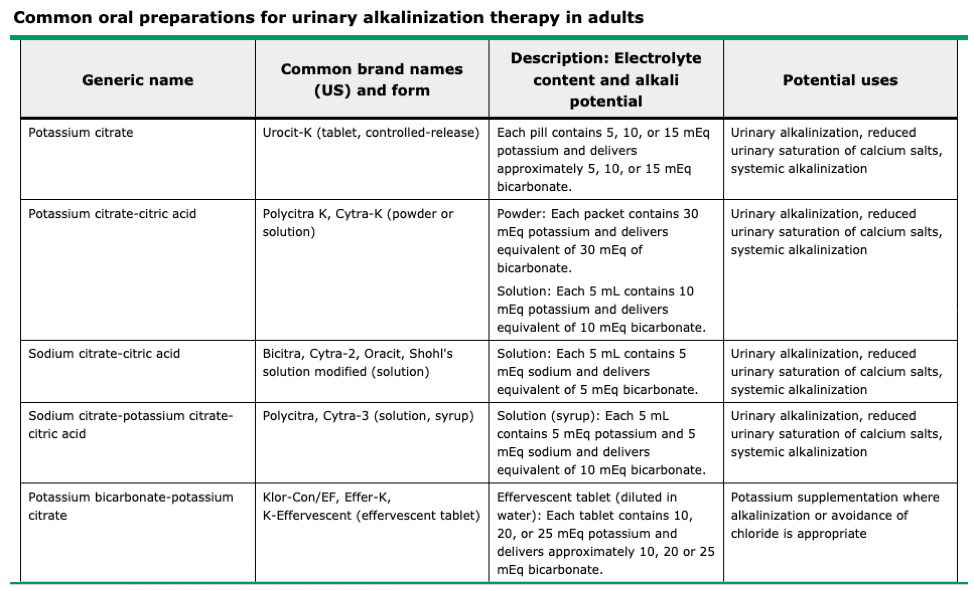
Treatment
- Type I (distal)
- Urinarary Alkali Therapy
- Sodium bicarbonate
- Increased risk of nephrolithiasis due
to bicarbonaturia
- Use potassium citrate instead
- Increased risk of nephrolithiasis due
to bicarbonaturia
- Sodium bicarbonate
- Urinarary Alkali Therapy
- Type II (proximal)
- Much more difficult to treat due to the INCREASED bicarbonate diuresis during bicarbonate therapy
- Alkali therapy (10x the dose for type I) AND potassium salt repletion as bicarbonaturia INCREASES urinary potassium losses
- Thiazide diuretics if large alkali
doses ineffective or not tolerated
- Diuresis reduces urinary bicarbonate
loss by increasing proximal sodium reabsorption
- Which secondarily increased bicarbonate reabsorption
- Diuresis reduces urinary bicarbonate
loss by increasing proximal sodium reabsorption
- Type IV
- Stop any medication causes or treat
underlying condition (hypoaldosteronism)
- Mineralcorticoid (fludrocortisone) and glucocorticoid (hydrocortisone)
- Potassium repletion
- Stop any medication causes or treat
underlying condition (hypoaldosteronism)
References
- Rodríguez Soriano J. Renal tubular acidosis: the clinical entity. Journal of the American Society of Nephrology : JASN. 2002; 13(8):2160-70. [pubmed]
- Skelton LA, Boron WF, Zhou Y. Acid-base transport by the renal proximal tubule. Journal of nephrology. ; 23 Suppl 16:S4-18. [pubmed]
- Hamm LL, Nakhoul N, Hering-Smith KS. Acid-Base Homeostasis. Clinical journal of the American Society of Nephrology : CJASN. 2015; 10(12):2232-42. [pubmed]
- The Curbsiders. Episode 104. https://thecurbsiders.com/internal-medicine-podcast/104-renal-tubular-acidosis-kidney-boy-joel-topf-md
- DB’s Medical Rants. http://www.medrants.com/archives/8897
- Oh M, Carroll HJ. Value and determinants of urine anion gap. Nephron. 2002; 90(3):252-5. [pubmed]
- Rodríguez Soriano J. Renal tubular acidosis: the clinical entity. Journal of the American Society of Nephrology : JASN. 2002; 13(8):2160-70. [pubmed]
- Karet FE. Mechanisms in hyperkalemic renal tubular acidosis. Journal of the American Society of Nephrology : JASN. 2009; 20(2):251-4. [pubmed]
PAINE #PANCE Pearl – Renal
Question
42yo woman, with a history systemic lupus erythematosus, presents to your clinic with a 1-month history of progressive leg swelling and polyuria. She is complaint with her medications and states that she hasn’t changed anything in her medical care. Physical examination reveals 2+ pitting edema to the knees in the lower extremities. BMP, UA, and urine microscopy are below.

- What is the next step in diagnosing this patient and what would you expect to find?