***LISTEN TO THE PODCAST HERE***
Anatomy and Physiology
- Primary components of the breast are terminal duct lobular ubnits, lobular stroma, interlobular stroma, ducts, and lactiferous sinuses
- Epithelium (terminal duct lobular units) is the most hormonally responsive
- Natural hormonal changes of puberty, pregnancy, lactation, and menopause can lead to remodeling of these structures

Main Classifications
- There are four main classifications of benign breast disorders that are based on the degree of cellular proliferations and atypia present
- Nonproliferative
- Characterized by acinar dilation and fibrosis
- Generally, not associated with increased risk of cancer
- Proliferative without atypia
- Characterized by accumulation of luminal epithelial cells
- Small increased risk of cancer (1.5-2x general population)
- Atypical hyperplasia
- Change in size, shape, or nuclear function of epithelial cells
- High risk of cancer development
- Miscellaneous
- Nonproliferative
Epidemiology
- 50% of women will experience a non-cancerous breast mass at some point in their lives
- Age of diagnosis
- Mean age of 51 years
- Younger in proliferative
- Older in atypical
- Mean age of 51 years
- Family History
- Strongest in patients with atypical
NONPROLIFERATIVE DISORDERS
Breast Cysts
- Most common
- 25% of nonproliferative
- 35-50 year olds
- Fluid-filled, round mass originating from the terminal duct lobular unity
- Patient Presentation
- Painful or painless
- Often solitary
- Physical Examination
- Smooth and firm to palpation with distinct border
- Diagnostic Studies
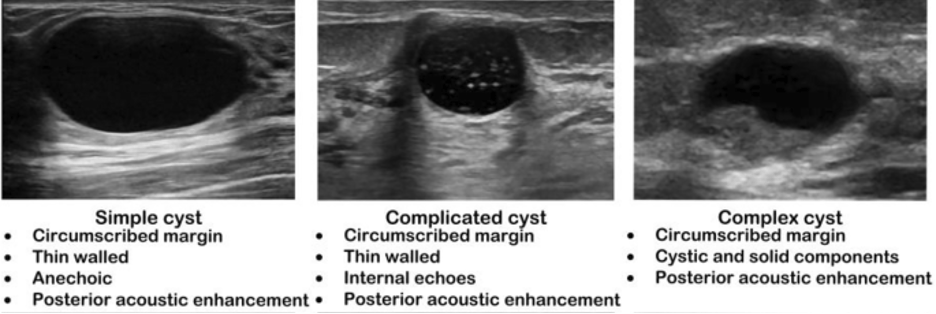
- Ultrasound
- Simple
- Anechoic throughout with posterior acoustic enhancement
- Complicated
- Homogenous low-level internal echoes with debris, thick walls, or thick septa
- No solid components
- Homogenous low-level internal echoes with debris, thick walls, or thick septa
- Complex
- Fluid and solid components without posterior wall enhancements
- Simple
- Ultrasound
- Management
- Simple – no intervention required
- Complicated – repeat imaging in 6 months
- Complex – biopsy or FNA
Galactocele
- Milk retention cysts usually caused by obstructed milk ducts
- Physical Examination
- Soft, cystic mass
- Diagnostic Studies
- Ultrasound
- Complex echogenicity
- Ultrasound

- Management
- FNA reveals milky substance
- No further intervention required
Hyperplasia of Usual Type
- Increase in the number of epithelial cells within a duct that is more than two, but not more than four cells in depth and do not cross the lumen of the involved space
PROLIFERATIVE DISORDERS WITHOUT ATYPIA
Fibroadenoma
- Most common benign tumor of the breast
- 50% of all breast biopsies
- 20% have multiple
- Most common in younger women
- 15-35 years of age
- Likely hormonally driven
- Persist through reproductive year’s, increase during pregnancy or with estrogen therapies, and decrease after menopause
- Physical Examination
- Well-defined, mobile mass on palpation
- Diagnostic Studies
- Ultrasound
- Well-defined solid mass with isoechogenicity
- Ultrasound

- Management
- Biopsy is indicated to further stratify
- Simple
- Contains glandular and fibrous tissue
- Watch vs excision vs cryoablation
- If any change during observation, then excision is warranted
- Complex
- Contains duct epithelial hyperplasia or calcification
- Observation vs excision
- Simple
- Biopsy is indicated to further stratify
Intraductal Papilloma
- Papillary cells that grown from the wall of a cyst into its lumen
- Can hide areas of atypia or ductal carcinoma in situ
- Two types
- Solitary
- Solid mass on examination or incidental imaging
- Nipple discharge is common presenting sign
- Multiple
- Minimum of five papillomas within a localized segment of tissue
- Solitary
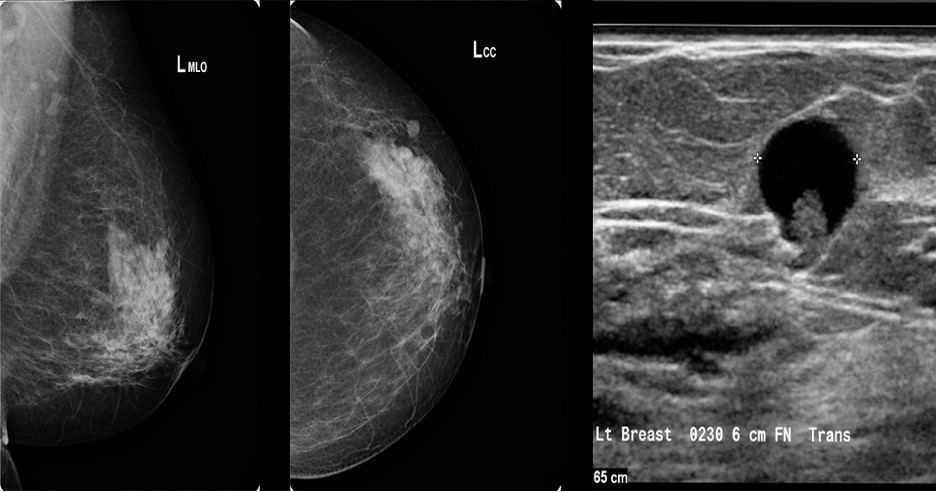
- Diagnostic Studies
- Often found on routine mammography, but ultrasound is recommended if there is a palpable mass
- Will show a mass within a cystic space
- Often found on routine mammography, but ultrasound is recommended if there is a palpable mass
- Management
- Solitary
- Biopsy and excision if atypical cells present
- Multiple
- Excision of breast segment
- Solitary
Sclerosing Adenosis
- Lobular lesions with increased fibrous tissue and interspersed glandular cells
- Found on routine mammography
- Architectural distortion with irregular borders and microcalcifications
- No interventions needed outside of routine imaging
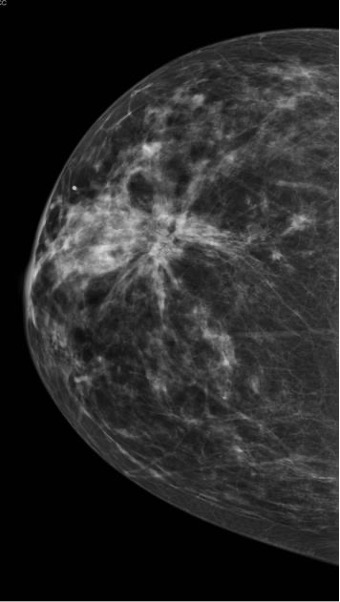
Radial Scar
- Complex sclerosing lesions found on routine imaging AFTER biopsies or excisions have been performed
- Pathologic by definition due to appearance
- Diagnostic Studies
- Mammography often shows low-intensity, spiculated masses that are indistinguishable from spiculated carcinomas
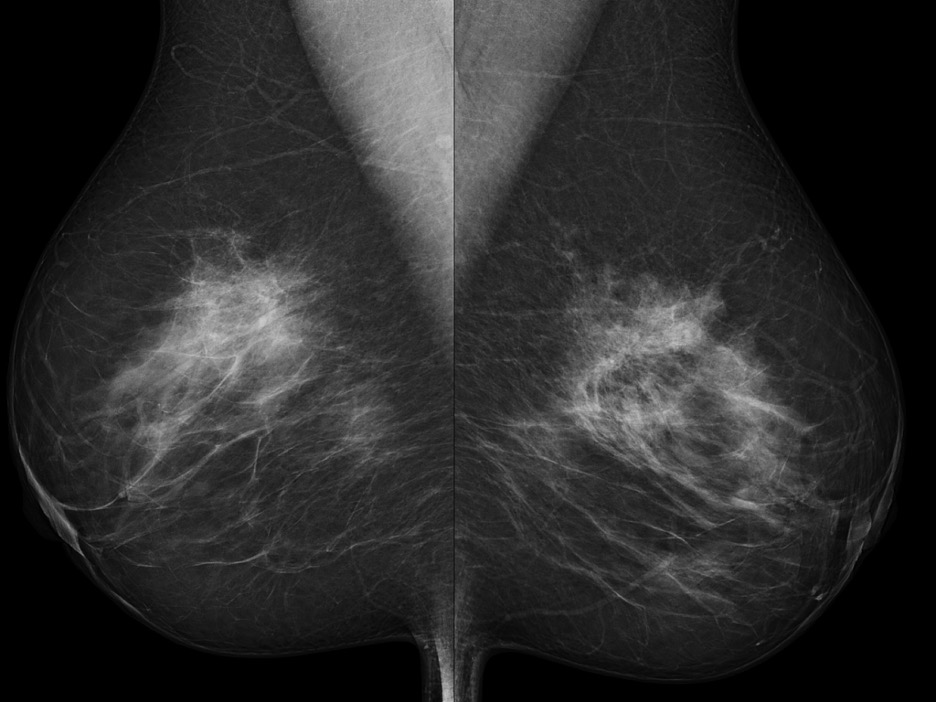
- Management
- Biopsy reveals fibroelastic cores with radiating ducts and lobules
- Excision is recommended (though controversial) and is often definitive
PROLIFERATIVE LESIONS WITH ATYPIA
Atypical Ductal Hyperplasia (ADH)
- Characterized by proliferation of uniform epithelial cells with monomorphic round nuclei filling the involved duct
- Must be < 2mm or involving < 2 ducts
- Can share cytologic and morphologic features of low-grade ductal carcinoma in-situ
- Diagnosed by core needle biopsy
- Management
- Excisional breast biopsy with good margins
Atypical Lobular Hyperplasia (ALH)
- Characterized by proliferation of monomorphic, evenly spaced, dyshesive cells filling the involved lobule
- Generally found on incidental biopsy for other clinical reason
- Can share cytologic and morphologic features of low-grade lobular carcinoma in-situ
- Diagnosed by core needle biopsy
- Management
- Excisional breast biopsy with good margins
Lobular Carcinoma in-situ
- Invasive lesion that arises from the lobules and terminal ducts of the breast
- 80-90% of cases diagnosed in premenopausal women with a mean age of 45 year’s
- Strong estrogen receptor positivity
- Diagnosed by core needle biopsy on other incidental reason
- LCIS is generally not diagnosed clinically, radiographically, or by gross pathologic examination
- Three types
- Classic
- Solid proliferation of small cells with small, uniform round nuclei and variably distinct cell borders with cytologic dyshesion
- Clear to lightly eosinophilic cytoplasm with possible signet ring cells and vacuoles
- Pleomorphic
- Larger cells with marked nuclear pleomorphism
- Florid
- Marked distension of the involved ducts and lobules that become mass-forming
- Central necrosis with calcifications
- Classic
- Management
- Excisional breast biopsy
Flat Epithelial Atypia
- Characterized by neoplastic alteration of the terminal duct lobular units with replacement of the native epithelial cells with columnar cells
- Diagnosed by core needle biopsy after mammographic evidence of microcalcifications
- Management
- Excisional breast biopsy
MISCELLANEOUS
Lipoma
- Solitary mature fat tumors of the breast that do not contain histologic evidence of breast tissue
- Physical Examination
- Soft, non-tender, well-circumscribed mass
- Difficulty to clinically differentiate from other conditions
- Soft, non-tender, well-circumscribed mass
- Excisional biopsy is preferred

Fat Necrosis
- Occurs as a result of breast trauma or surgical intervention
- Can be confused with malignancy both clinically and radiographically
- May see oil cysts on mammography or ultrasound
- Biopsy is often needed to diagnose, but no further treatment is indicated

Diabetic Mastopathy
- Seen in premenopausal women with long standing type 1 diabetes mellitus
- Mammogram shows dense pattern
- Diagnosed by core needle biopsy
- Shows dense, keloid-like fibrosis with periductal, lobular, or perivascular lymphocytic infiltration
- No further treatment after diagnosis
Hamartoma
- Lesions containing varying amounts of glandular, adipose, or fibrous tissue
- Present as discrete, encapsulated, painless masses found on incidental radiographic screening
- FNA or CNB are not sufficient to make the diagnosis and excisional biopsy is preferred
1893 Cottage Physician


References
- https://armandoh.org/disease/breast-cancer/
- Schnitt SJ. Benign breast disease and breast cancer risk: morphology and beyond. Am J Surg Pathol. 2003; 27(6):836-41. [pubmed]
- Hartmann LC, Sellers TA, Frost MH, et al. Benign breast disease and the risk of breast cancer. N Engl J Med. 2005; 353(3):229-37. [pubmed]
- Guray M, Sahin AA. Benign breast diseases: classification, diagnosis, and management. Oncologist. 2006; 11(5):435-49. [pubmed]
- Breast Disease. In: Hoffman BL, Schorge JO, Halvorson LM, Hamid CA, Corton MM, Schaffer JI. eds. Williams Gynecology, 4e. McGraw-Hill; Accessed February 21, 2021. https://accessmedicine-mhmedical-com.ezproxy.uthsc.edu/content.aspx?bookid=2658§ionid=218608871
- Giuliano AE, Hurvitz SA. Breast Disorders. In: Doherty GM. eds. Current Diagnosis & Treatment: Surgery, 15e. McGraw-Hill; Accessed February 21, 2021. https://accessmedicine-mhmedical-com.ezproxy.uthsc.edu/content.aspx?bookid=2859§ionid=242155824
- Littrup PJ, Freeman-Gibb L, Andea A, et al. Cryotherapy for breast fibroadenomas. Radiology. 2005; 234(1):63-72. [pubmed]
- Linda A, Zuiani C, Furlan A, et al. Radial scars without atypia diagnosed at imaging-guided needle biopsy: how often is associated malignancy found at subsequent surgical excision, and do mammography and sonography predict which lesions are malignant? AJR Am J Roentgenol. 2010; 194(4):1146-51. [pubmed]
- American Society of Breast Surgeons. Official statement. Consensus guideline on concordance assessment of image-guided breast biopsies and management of borderline or high-risk lesions. 2016. Available at: https://www.breastsurgeons.org/docs/statements/Consensus-Guideline-on-Concordance-Assessment-of-Image-Guided-Breast-Biopsies.pdf
- Guray M, Sahin AA. Benign breast diseases: classification, diagnosis, and management. Oncologist. 2006; 11(5):435-49. [pubmed]