***LISTEN TO THE PODCAST HERE***
Click Here for Episode #11 – Review of Cyanotic Congenital Heart Defects
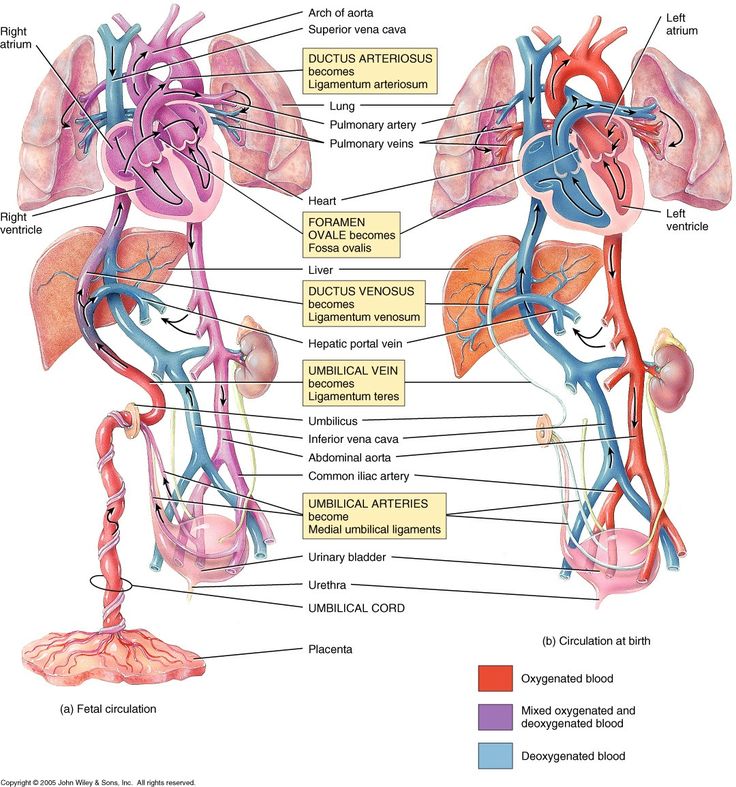
Review of In-Utero and Neonatal Cardiovascular Physiology
- Ductus arteriosus
- Connects the pulmonary artery to the descending aorta
- Prostaglandin E2 are produced by the placenta and keep this open, along with low arterial oxygen concentration
- Begins to close 10-15 hours after delivery and should be completed by 2-3 weeks of age
- Foramen ovale
- Communication between right and left atrium
- Once the infant begins spontaneously breathing, increases in pulmonary blood flow and left atrial pressures mechanically seals the foramen ovale
Review of Initial Approach to Screening for Congenital Heart Diseases
- The main approach to initial cardiac evaluation for infants with suspected heart disease is to determine:
- Innocent vs Congenital Heart Disease
- Cyanotic vs Acyanotic
- Indications for emergent referral
- Innocent vs Congenital Heart Disease
- Historical Clues
- Symptomatic
- Cyanosis, respiratory, poor growth, poor feeding, syncopal episodes
- Family History
- Parent or sibling with congenital heart disease
- 1st degree relative with any CHD – RR 3.21
- Parent or sibling with congenital heart disease
- Genetic syndromes
- Prenatal Evaluation
- Abnormalities on prenatal ultrasound
- Maternal factors that increase risk
- Multifetal pregnancy, prematurity, preeclampsia, DM, HTN, Age > 40, alcohol/substance use, smoking,
- Age of child
- Symptomatic
- Physical Examination
- Murmurs
- Innocent murmur
- ≤ Grade 2
- Short systolic phase
- Minimal radiation
- Soft intensity
- Innocent murmur
- Extra Heart Sounds
- Abnormal S2
- S3 or S4
- Systolic click
- Thrills and heaves
- Other Findings
- Abnormal vital signs
- BP differences between right arm, left arm, and legs
- Weak or bounding pulses
- Hepatomegaly
- Abnormal vital signs
- Murmurs
- Diagnostic Studies
- Pulse oximetry screening
- Measure in right hand (pre-ductal) and either foot (post-ductal)
- Chest radiograph
- Cardiomegaly, increased pulmonary vascular markings, pulmonary edema
- EKG
- LVH, RVH, abnormal axis, dysrhythmias, prolonged QT
- Echocardiogram
- Pulse oximetry screening
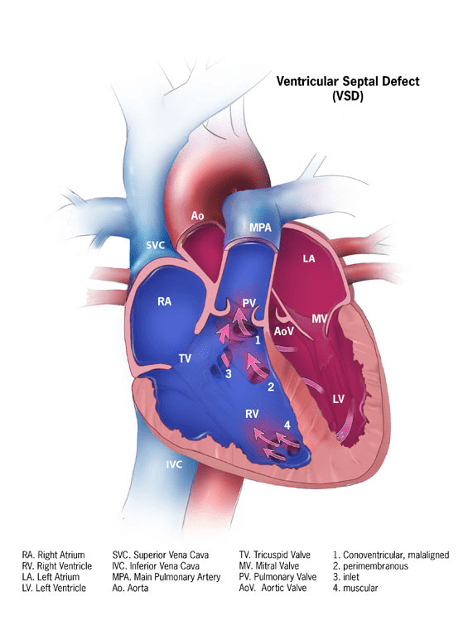
Ventricular Septal Defect
Epidemiology
- 30% of all CHD (most common)
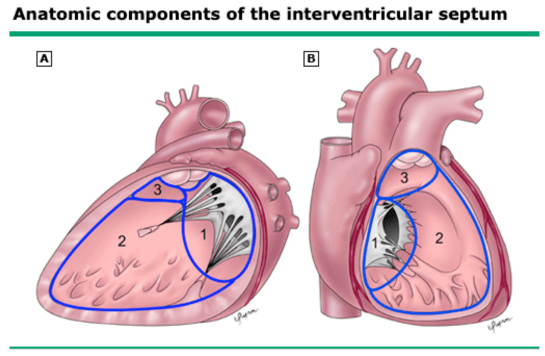
Normal Development
- Three main components
- AV canal septum (1)
- Muscular septum (2)
- Parietal band or distal conal septum (3)
- Closure of the interventricular foramen is dependent on:
- Right-sided endocardial cushions projecting into the AV canal
- Connective tissue growth on the crest of the muscular septum
- Downward growth of ridges dividing the conus
Types
- Membranous Defects
- Most common clinically significant VSD
- Under aortic valve and behind septal leaflet of the tricuspid valve
- May extend into muscular septum
- Can be associated with LVOTO and coarctation
- Muscular Defects
- Located along right ventricular free wall-septal junction, in the central muscular septum, or in the apical septum
- Often close spontaneously
- Malalignment Defects
- Result from anterior/posterior malalignment of the conal septum
- Associated with Tetralogy of Fallot
- Subpulmonic Defects (outlet)
- Superior and anterior conal septum defects
- AV Canal Defects (inlet)
- Posterior and superior to annulus of tricuspid valve
- Associated with ASDs

Pathophysiology
- Cause no problems in-utero
- Causes a left-to-right shunt ex-utero
- Higher left ventricular pressures
- Categorization is based on:
- Size
- Small – < 4mm
- Moderate – 4-6mm
- Large – > 6mm
- Shunt (Pulmonary:Systemic flow)
- Small – Qp:Qs < 1.5
- Moderate – Qp:Q 1.5-2.3
- Large – Qp:Qs > 2.3
- Size
- Effects on Circulation
- Pulmonary
- Increased and causes tachypnea and increased respiratory effort
- Systemic
- LV output must increase to maintain systemic flow
- As systemic flow decreases (large VSDs):
- Increased alpha-adrenergic stimulation
- Increased catecholamine release
- Increased angiotensin release
- Increases risk of heart failure
- Pulmonary
Natural History
- Small
- 75% close spontaneously within 2 years of life
- Those that persist in adulthood are benign
- Moderate
- Spontaneous closure depends on pulmonary arterial pressure and size/location of defect
- Can respond to medical management
- Large
- Rarely spontaneously close
- Surgery must be performed within 1st year to avoid permanent pulmonary vascular resistance
Clinical Presentation
- Prenatal
- Moderate to large VSD can be diagnosed during 20week ultrasound
- Postnatal
- Small VSD
- Can be asymptomatic and present with only a murmur
- Moderate to Large VSD
- Present 3-4 weeks from birth with signs and symptoms of heart failure
- Tachypnea
- Poor feeding or poor weight gain
- Tachycardia
- Hepatomegaly
- Rales, retractions
- Cardiac Examination
- Murmur
- Harsh or blowing holosystolic
- Heard best at 3rd-4th intercostal space
- May have a diastolic rumble
- Increased flow across mitral valve
- Loud, splitting of S2
- Due to increased pulmonary arterial pressure
- Palpable thrill may be present
- Murmur
- Present 3-4 weeks from birth with signs and symptoms of heart failure
- Small VSD
Diagnostic Studies
- EKG
- LVH, RVH, RAE
- Chest Radiograph
- Increased pulmonary vascular markings
- Echocardiography
- Two-dimensional Doppler confirms diagnosis
Management
- Expectant
- Most small VSD close spontaneously
- Follow-up every 6 months with cardiologist until murmur resolves or yearly if murmur persists but still asymptomatic
- Medical
- Heart failure management
- Diuretics are first line
- Nutritional support
- Pulmonary hypertension management
- May need cardiac catheterization for accurate measurements and determination for surgical management
- Heart failure management
- Surgical
- Indications
- Persistent symptoms with maximal medical therapy
- Moderate/large defects with pulmonary hypertension
- Persistent left-to-right shunt with LV dilation
- Associated aortic valve prolapse or aortic regurgitation
- Double-chambered right ventricle
- Direct patch closure is procedure of choice in most children
- Transcatheter closure is technically challenging and not offered routinely due to higher incidence of AV block and valve injury
- Indications
Atrial Septal Defect
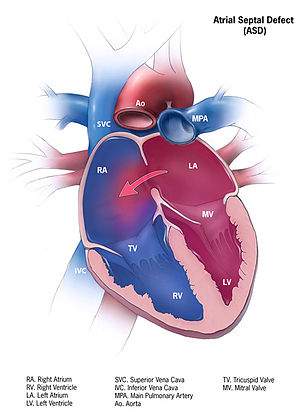
Epidemiology
- 10-15% of all CHD
- 1-2 per 1000 live births
Normal Development
- Begins at 5th week and is made up of 3 structures
- Septum primum and AV canal septum (endocardial cushions)
- Arises from superior portion of common atrium
- Grows caudally towards AV canal septum
- These two fuse close ostium primum between right and left atria
- Septum secundum
- Covers the ostium secundum on the right atrial side of the septum primum
- Forms the fossa ovalis
- Leaves a small opening in-utero
- Foramen ovale
- Held open due to pressure gradients between the higher right atria and lower left atria
- Allows for right to left flow
- Held open due to pressure gradients between the higher right atria and lower left atria
- Foramen ovale
- Covers the ostium secundum on the right atrial side of the septum primum
- Septum primum and AV canal septum (endocardial cushions)
Types
- Primum Defect
- 15-20% of ASDs
- Occurs when primum septum does not fuse with endocardial cushions
- Secundum Defect
- 70% of ASDs
- 2x more common in females
- Located within the fossa ovalis
- Sinus Venosus Defect
- 5-10% of ASDs
- Malposition of the insertion of the superior or inferior vena cava on the atrial septum
- Patent Foramen Ovale
- Identified on 30% of adult autopsies
- NOT considered an ASD because no septal tissue is missing
Natural History
- Most close spontaneously
- >80% of small (<5mm)
- 30-60% of moderate (6-10mm)
- Large (>10mm) do not close spontaneously
- Persistent ASDs can cause pulmonary hypertension and heart failure in adulthood
Clinical Presentation
- Most are asymptomatic and diagnosed only by identification of murmur
- Murmur
- Midsystolic pulmonary flow or ejection murmur
- Heard best left 2nd intercostal space
- Wide, fixed split S2
- ASD equalizes the respiratory effect on both right and left ventricular output
- If pulmonary hypertension is present, there may be an accentuated pulmonic component of S2
- May have associated mitral regurgitation murmur
- Midsystolic pulmonary flow or ejection murmur
Diagnostic Studies
- EKG
- May show rSr’ or rsR’ pattern in V1
- Incomplete RBBB
- May show rSr’ or rsR’ pattern in V1
- Echocardiogram
- Transthoracic may see, but a transesophageal is better
Management
- Often watch until child is 2yo as most will spontaneous
- Even persistent defects are not recommended to close
- Closure Indications
- Right heart enlargement
- Pulmonary overcirculation
- Substantial left-to-right shunt
- Types of Closures
- Percutaneous
- Criteria
- < 30mm is diameter
- ≥ 5mm or rim tissue around defect sufficient for effective closure without obstructing surrounding structures
- Avoids bypass and major surgical incisions
- Complications
- Device embolization, malposition, dysrhythmias, cardiac perforation
- Criteria
- Surgery
- Indications
- Sinus venosus defects
- Coronary sinus defects
- Primum ASDs
- Large ASDs with heart failure
- Closed with pericardial or Dacron patch
- Indications
- Percutaneous
Atrioventricular Septal Defect
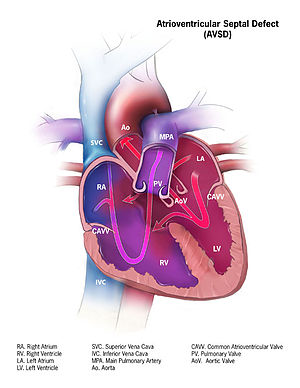
Epidemiology
- 4-5% of CHD
- 0.3-0.4 per 1000 live births
- Up to a 50% risk of trisomy 21
Normal Development
- Primitive AV canal connects the atria with the ventricles
- At 4-5 weeks gestation, the superior and inferior endocardial cushions fuse and form the AV canal
- This contributes to the AV valves and septum
Types and Classifications
- Classified based on the degree of the defect
- Complete
- Complete failure of fusion between the superior and inferior endocardial cushions
- Combined primum ASD and posterior VSD with a single, common AV valve
- Can be balanced (both ventricles get same flow) or unbalanced (one gets more than the other)
- Unbalanced can lead to hypoplasia
- Complete failure of fusion between the superior and inferior endocardial cushions
- Partial
- Incomplete fusion of superior and inferior endocardial cushions
- Primum ASD, single AV valve annulus with 2 valve orifices
- Incomplete fusion of superior and inferior endocardial cushions
- Transitional
- Large primum defect, cleft mitral valve, and inlet VSD
- Complete
- Rastelli Classification
- Type A
- Most common form and most common ASVD with Down syndrome
- Type B
- Least common
- Type C
- Frequently found with other conditions
- Tetralogy of Fallot, Transposition of Great Vessels
- Frequently found with other conditions
- Type A
Pathophysiology
- Complete AV Canal Defect
- Increased pulmonary blood flow due to left-to-right shunting
- Leads to heart failure and pulmonary hypertension
- Increased pulmonary blood flow due to left-to-right shunting
- Partial AV Canal Defect
- Volume overload of right atrium and right ventricle with pulmonary overcirculation due to left-to-right shunting
- No pulmonary hypertension
- Mitral regurgitation can be severe
- Volume overload of right atrium and right ventricle with pulmonary overcirculation due to left-to-right shunting
- Transitional AV Canal Defect
- Shunting often mild due to small VSD
Clinical Presentation
- In-Utero
- Can be diagnosed early during pregnancy
- Rarely cause any fetal distress or growth disturbances
- Complete
- Heart failure develops early in infancy
- Tachypnea, poor feeding, poor growth, sweating, and pallor by 2 month
- Severity of symptoms depends on size and degree of AV valve regurgitation
- Physical examination
- Hyperactive precordium with inferior/laterally displaced PMI
- Increased S2
- Systolic ejection murmur heard best at left upper sternal border
- Heart failure develops early in infancy
- Partial and Transitional
- Asymptomatic during childhood and only diagnosed on routine examination
- Physical examination
- Wide and fixed S2 during respiration
- Systolic ejection murmur heard best at left upper sternal border
- Diastolic rumble may be heard
- Holosytolic murmur of MR
Diagnostic Studies
- Echocardiography
- Transthoracic is adequate
- Apical four chamber or subcostal view is best to see defect
Management
- In general, surgical correction is recommended because of significant morbidity and mortality of the pulmonary circulation effects
- Complete
- Correction by 6 months of age to prevent permanent pulmonary hypertension
- Close the ASD and VSD and create to separate, competent AV valves
- Single-patch or double-patch
- Partial and Transitional
- Primum ASD does not close spontaneously and leaves the patient at risk for atrial fibrillation and heart failure later in life
- Surgical goal is to close intraatrial communication and restore/preserve AV valve competence
- Patch closure of ostium primum defect and mitral valvuloplasty
- Timing is usually between 18 months and 3 years
Patent Ductus Arteriosus

Epidemiology
- 3-8 per 10,000 live births
- 2:1 female predominance
Normal Development
- Derived from the embryonic left sixth arch
- Actually has different tissue than the aorta or pulmonary artery
- Intima is thicker
- Media contains more smooth muscle
- In utero, allows from RV flow to placenta to get oxygenated
- Keep open by low arterial oxygen and prostaglandin E2 from placenta
- At birth, increased levels of arterial oxygen and the lack of prostaglandin E2 begins the process of spontaneous closure
Clinical Presentation
- Symptoms depend on the degree of left-to-right shunting
- Dependent on size and length of PDA, as well as the difference between the pulmonary and systemic vascular resistances
- Classic murmur
- Continuous, holosystolic murmur
- Machinery murmur
- Heard best over left upper sternal border
- Continuous, holosystolic murmur
- Small (Qp:Qs < 1.5 to 1)
- Asymptomatic and detected on routine physical examination
- Moderate (Qp:Qs 1.5-2.2 to 1)
- May present with exercise intolerance and heart failure
- Left-to-right shunt increasing left atrial and ventricular volume
- Progress to left ventricular dilation and dysfunction
- Left-to-right shunt increasing left atrial and ventricular volume
- Displaced PMI
- Widened pulse pressure
- May present with exercise intolerance and heart failure
- Large (Qp:Qs > 2.2 to 1)
- Due to increased left sided pressures, can lead to increased pulmonary pressures
- May be irreversible if not corrected
- Can lead to right-to-left shunt if pressure is high enough
- Eisenmenger syndrome (cyanosis)
- Split S2 with prominent pulmonary component
- Murmur may disappear as pulmonary pressure increases
- Heart failure, poor feeding, failure to thrive, respiratory distress
- Dynamic PMI with a thrill
- Wide pulse pressures and bounding pulses
- Due to increased left sided pressures, can lead to increased pulmonary pressures
Diagnostic Studies
- Transthoracic, doppler color flow echocardiography
- The ductus is best viewed on the parasternal short-axis and suprasternal view
Management
- Decisions are made on whether to actively close the PDA or monitor cardiac status
- Term vs pre-term
- Pre-term respond well to prostaglandin inhibitors
- Size of the PDA
- Moderate to large warrant closure
- Small, audible PDA also benefit from closure to lower long term complications
- Especially bacterial endocarditis
- Degree of left-to-right shunt
- Degree of left sided volume overload
- Evidence of pulmonary hypertension
- Closure not recommended in severe PAH
- Term vs pre-term
- Closure Options
- Medical/Pharmacologic Therapy
- Prostaglandin inhibitors
- Indomethacin, ibuprofen
- Ibuprofen > indomethacin in term infants and older patients
- Indomethacin, ibuprofen
- Prostaglandin inhibitors
- Surgery
- Posterolateral thoracotomy with direct PDA ligation
- Video-Assisted Thoracoscopic Surgery (VATS) is less invasive option
- Not indicated if PDA size > 9mm
- Percutaneous closure using coils or commercial occlusion devices
- Medical/Pharmacologic Therapy
Coarctation of the Aorta

Definition
- Narrowing of the descending aorta at the insertion of the ductus arteriosus distal to the left subclavian artery
Epidemiology
- 4-6% of all CHD
- 4 cases per 10,000 live births
- More common in males
Pathogenesis
- Unknown, but two theories prevail:
- Decreased intrauterine blood flow leading to under development of the fetal aortic arch
- Migration/extension of ductal tissue into the wall of thoracic aorta
- Genetic associations and increased familial risk
- Usually accompanied with other CHD
Pathophysiology
- As PDA and FO begin to close after birth, increased pressure over the narrowed aorta
- Leads to left ventricular outflow tract obstruction
- Increased collateral blood flow across the intercostals, internal mammary, and scapular vessels
Clinical Manifestations
- Classic findings
- Absent/delayed femoral pulses
- Difference in upper extremity and lower extremity blood pressures
- Neonates
- Asymptomatic as long as there is a patent ductus
- Murmurs of other CHD
- Infants and Older Children
- Complain of lower extremity claudication symptoms with exertion
- Hypertension may be prominent
- Adults
- Hypertension is may sign
- If severe, may lead to heart failure, aortic pathologies
- Claudication can also be present
- Hypertension is may sign
Diagnostic Studies
- Chest radiography
- Infants
- Cardiomegaly with increased pulmonary vascular markings
- Older children and adults
- Rib notching from large collateral development
- “3” sign from indentation of aortic wall at coarctation site
- Infants

- Echocardiography
- Transthoracic, doppler flow can visualize coarctation and evaluate for other defects
- Ideal view is high parasternal long axis or suprasternal view
- Cardiovascular MRI/CT
- Recommended for adolescent and adult patients
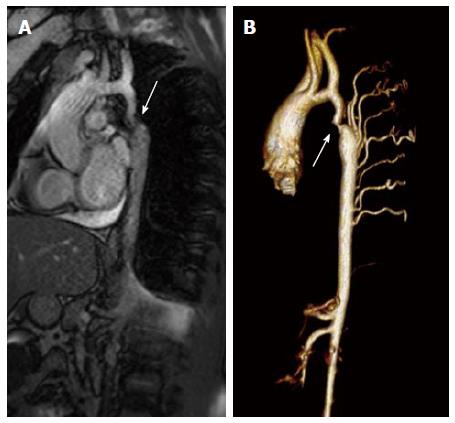
- Cardiac catheterization
- Generally performed in conjunction with therapeutic interventions, or in adults with associated coronary disease
Management
- Indications for inventions
- Neonates with critical coarctation
- Emergent medical therapy
- Continuous infusion of prostaglandin E1
- Inotropic support
- Support care to correct metabolic derangements
- Emergent medical therapy
- Gradient > 20 mmHg
- Radiologic evidence of clinically significant collateral flow
- Hypertension or heart failure attributed to the defect
- Neonates with critical coarctation
- Types of Interventions
- Balloon angioplasty (only)
- Infants > 4 months and young children < 25kg
- Preferred therapy for isolated coarctation
- Stent Placement (after angioplasty)
- Children > 25kg and adults
- Surgical repair
- Falling out of favor with increasing advancement and safety of transcatheter techniques
- Balloon angioplasty (only)
References
- Liu S, Joseph KS, Lisonkova S, et al. Association between maternal chronic conditions and congenital heart defects: a population-based cohort study. Circulation. 2013; 128(6):583-9. [pubmed]
- Jenkins KJ, Correa A, Feinstein JA, et al. Noninherited risk factors and congenital cardiovascular defects: current knowledge: a scientific statement from the American Heart Association Council on Cardiovascular Disease in the Young: endorsed by the American Academy of Pediatrics. Circulation. 2007; 115(23):2995-3014. [pubmed]
- Øyen N, Poulsen G, Boyd HA, Wohlfahrt J, Jensen PK, Melbye M. Recurrence of congenital heart defects in families. Circulation. 2009; 120(4):295-301. [pubmed]
- Frank JE, Jacobe KM. Evaluation and management of heart murmurs in children. American family physician. 2011; 84(7):793-800. [pubmed]
- Kang G, Xiao J, Wang Y, et al. Prevalence and clinical significance of cardiac murmurs in schoolchildren. Archives of disease in childhood. 2015; 100(11):1028-31. [pubmed]
- McCrindle BW, Shaffer KM, Kan JS, Zahka KG, Rowe SA, Kidd L. Cardinal clinical signs in the differentiation of heart murmurs in children. Archives of pediatrics & adolescent medicine. 1996; 150(2):169-74. [pubmed]
- van der Linde D, Konings EE, Slager MA, et al. Birth prevalence of congenital heart disease worldwide: a systematic review and meta-analysis. Journal of the American College of Cardiology. 2011; 58(21):2241-7. [pubmed]
- Hoffman JI, Kaplan S. The incidence of congenital heart disease. Journal of the American College of Cardiology. 2002; 39(12):1890-900. [pubmed]
- Zhao QM, Niu C, Liu F, Wu L, Ma XJ, Huang GY. Spontaneous Closure Rates of Ventricular Septal Defects (6,750 Consecutive Neonates). The American journal of cardiology. 2019; 124(4):613-617. [pubmed]
- Zhang J, Ko JM, Guileyardo JM, Roberts WC. A review of spontaneous closure of ventricular septal defect. Proceedings (Baylor University. Medical Center). 2015; 28(4):516-20. [pubmed]
- Bol-Raap G, Weerheim J, Kappetein AP, Witsenburg M, Bogers AJ. Follow-up after surgical closure of congenital ventricular septal defect. European journal of cardio-thoracic surgery : official journal of the European Association for Cardio-thoracic Surgery. 2003; 24(4):511-5. [pubmed]
- Butera G, Carminati M, Chessa M, et al. Transcatheter closure of perimembranous ventricular septal defects: early and long-term results. Journal of the American College of Cardiology. 2007; 50(12):1189-95. [pubmed]
- Hanslik A, Pospisil U, Salzer-Muhar U, Greber-Platzer S, Male C. Predictors of spontaneous closure of isolated secundum atrial septal defect in children: a longitudinal study. Pediatrics. 2006; 118(4):1560-5. [pubmed]
- Muta H, Akagi T, Egami K, et al. Incidence and clinical features of asymptomatic atrial septal defect in school children diagnosed by heart disease screening. Circulation journal : official journal of the Japanese Circulation Society. 2003; 67(2):112-5. [pubmed]
- Korenberg JR, Bradley C, Disteche CM. Down syndrome: molecular mapping of the congenital heart disease and duodenal stenosis. American journal of human genetics. 1992; 50(2):294-302. [pubmed]
- Cetta F, Minich LL, Edwards WD, et al. Atrioventricular septal defects. In: Moss and Adams’ Heart Disease in Infants, Children, and Adolescents Including the Fetus and Young Adult, 7th ed, Allen HD, Shaddy RE, Driscoll DJ, Feltes TF (Eds), Lippincott Williams & Wilkins, Philadelphia 2007.
- Rastelli G, Kirklin JW, Titus JL. Anatomic observations on complete form of persistent common atrioventricular canal with special reference to atrioventricular valves. Mayo Clinic proceedings. 1966; 41(5):296-308. [pubmed]
- Backer CL, Stewart RD, Mavroudis C. What is the best technique for repair of complete atrioventricular canal? Seminars in thoracic and cardiovascular surgery. 2007; 19(3):249-57. [pubmed]
- Minich LL, Atz AM, Colan SD, et al. Partial and transitional atrioventricular septal defect outcomes. The Annals of thoracic surgery. 2010; 89(2):530-6. [pubmed]
- Hoffman JI, Kaplan S. The incidence of congenital heart disease. Journal of the American College of Cardiology. 2002; 39(12):1890-900. [pubmed]
- Warnes CA, Williams RG, Bashore TM, et al. ACC/AHA 2008 Guidelines for the Management of Adults with Congenital Heart Disease: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (writing committee to develop guidelines on the management of adults with congenital heart disease). Circulation. 2008; 118(23):e714-833. [pubmed]
- Giroud JM, Jacobs JP. Evolution of strategies for management of the patent arterial duct. Cardiology in the young. 2007; 17 Suppl 2:68-74. [pubmed]
- Keane JF, Flyer DC. Coarctation of the aorta. In: Nadas’ Pediatric Cardiology, 2nd ed, Keane JF, Lock JE, Fyler DC (Eds), Saunders Elsevier, Philadelphia 2006.
- Beekman RH III. Coarctation of the Aorta. In: Moss and Adams’ Heart Disease in Infants, Children, and Adolescents, 6th ed, Allen HD, Driscoll DJ, Shaddy RE, Feltes TF (Eds), WK Lippincott Willams and Wilkins, Philadelphia 2008. Vol 2
- Silversides CK, Kiess M, Beauchesne L, et al. Canadian Cardiovascular Society 2009 Consensus Conference on the management of adults with congenital heart disease: outflow tract obstruction, coarctation of the aorta, tetralogy of Fallot, Ebstein anomaly and Marfan’s syndrome. The Canadian journal of cardiology. 2010; 26(3):e80-97. [pubmed]
- Baumgartner H, Bonhoeffer P, De Groot NM, et al. ESC Guidelines for the management of grown-up congenital heart disease (new version 2010). European heart journal. 2010; 31(23):2915-57. [pubmed]








Pingback: #70 – Newborn Examination | PAINE Podcast and Medical Blog