*** LISTEN TO THE PODCAST HERE***
Definition of Heart Failure
- Complex clinical syndrome resulting
from conditions that affect the structure and/or function of the heart
culminating in reduced systemic perfusion that is inadequate to meet the
metabolic demands of the body
- Main effect is decreased cardiac output
- No more “congestive”
Epidemiology
- 5.7 million adults in US
- 550,000 new cases each year
- 1.4 million are under 60 years of age
- Annual incidence is 10 per 1,000 population AFTER 65 years of age
- 287,000 deaths per year
- 1 in 9 deaths included heart failure as contributing causes
- Most common diagnosis in hospital patients over 65 years of age
- Responsible for 11 million office visits each year in the US and more hospitalizations than all cancers COMBINED
- 50% of adults who develop heart failure die within 5 years
- Cost to US is ~$30 billion/year

Causes and Pathophysiology
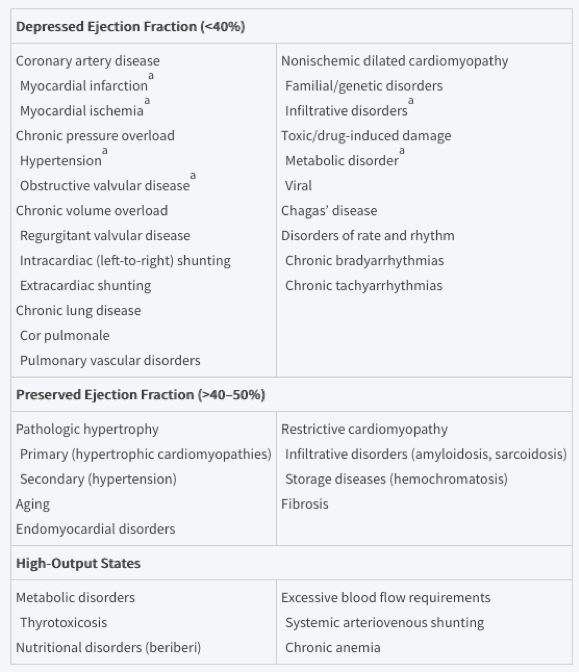
Any condition that leads to alteration in left ventricular structure or function can cause heart failure and the specific causes depends on the preservation of ejection fraction. There is considerable overlap between these with coronary artery disease and hypertension causes the majority of cases.
The problem is that the rest of the body feel the effects of the decreased cardiac output and activate the neurohormonal systems to compensate. The issue is that this makes the heart failure worse and it is a vicious cycle until it can be broken.


Signs and Symptoms
- History
- Reduced
cardiac output
- Fatigue, weakness
- Excessive
fluid accumulation
- Dyspnea, orthopnea, paroxysmal nocturnal dyspnea, leg swelling, abdominal discomfort, palpitations
- Reduced
cardiac output
- Physical
Examination
- Appearance
and Vital Signs
- Resting sinus tachycardia
- Narrow pulse pressure
- Cool, pale skin (peripheral vasoconstriction)
- Volume
Assessment
- Pulmonary
congestion
- Rales on auscultation
- AUDIO
- Peripheral
edema
- Leg swelling, hepatic congestion, ascites, scrotal edema
- Elevated
jugular venous pressure
- Hepatojugular reflux
- Pulmonary
congestion
- Appearance
and Vital Signs
- Cardiac
- S3 with gallop (if systolic failure)
- S4 (if diastolic failure)
- Displaced PMI past midclavicular line and below the 5th intercostal space
- Pulsus alternans
- Evenly spaced alternating strong and weak peripheral pulses

Framingham Clinical Criteria for Heart Failure

Clinical Decision Rule for Heart Failure
- Age
- < 60 = 0 points
- 60-70 = 4 point
- 71-80 = 7 points
- > 80 = 10 points
- History
of coronary disease = 15 points
- AMI, CABG, PCI
- Loop diuretic = 10 points
- Displaced PMI = 20 points
- Rales = 14 points
- Irregularly irregular pulse = 11 points
- Heart murmur = 10 points
- Pulse Rate = (HR-60)/3 points
- Elevated jugular venous pressure = 12 points
- NT-proBNP
(pg/mL)
- < 100 = 0 points
- 100-200 = 8 points
- 200-400 = 16 points
- 400-800 = 24 points
- 800-1600 = 32 points
- 1600-3200 = 40 points
- > 3200 = 48 points
- Interpretation
- < 13 points = < 10% probability of heart failure
- > 54 points = > 70% probability of heart failure
Diagnostic Studies
- Electrocardiogram
- Not really diagnostic, but can evaluate for current ischemia, past infarction, low voltage, dysrhythmias
- A normal EKG makes systolic dysfunction unlikely (98% NPV)
- Laboratory
studies
- Brain
natriuretic peptide (BNP) and N-terminal proBNP (NT-proBNP)
- Released from ventricles when stretched
- < 100 pg/mL = very high NPV and rule out heart failure
- Cardiac enzymes
- CBC
- CMP
- Brain
natriuretic peptide (BNP) and N-terminal proBNP (NT-proBNP)
- Chest
Radiography
- Increased cardiothoracic ratio
- Cephalization of pulmonary vessels
- Kerley B-lines
- Pleural effusions

- Echocardiography
- Recommended for all patient with dyspnea and suspicion of heart failure
- Provides vital information on:
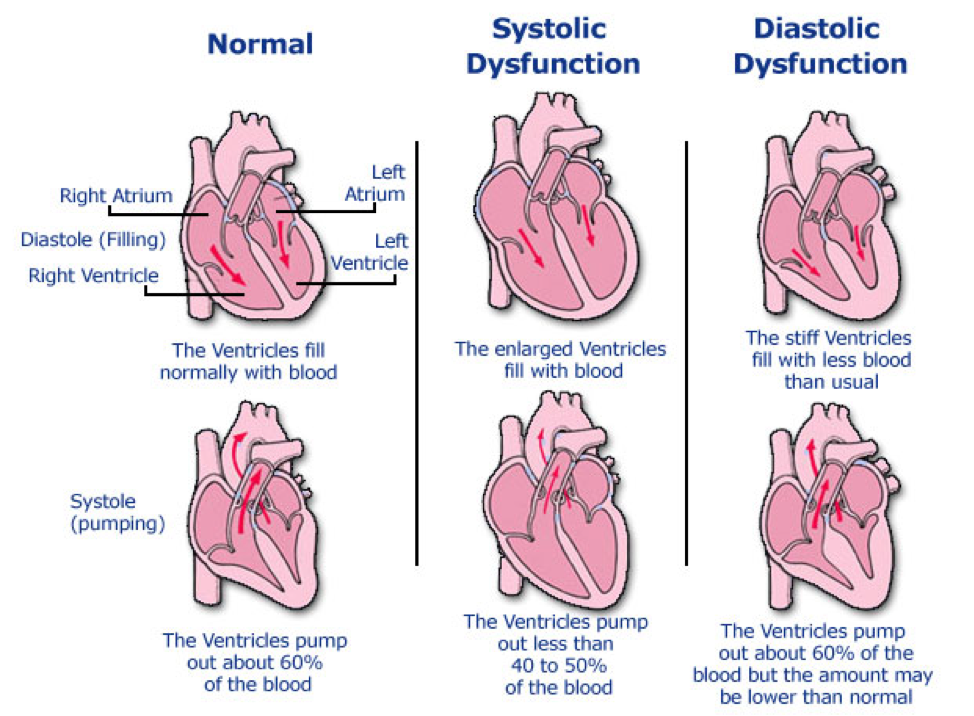
- Ejection fraction
- < 40% = reduced = systolic
- > 50% = preserved = diastolic
- Valvular disease
- Aortic and mitral regurgitation/insufficiency
- Atrial and Ventricular size and function
- Enlarged left ventricle = systolic
- Left atrial enlargement with normal/small left ventricle = diastolic
- Left ventricular wall size
- Thin = systolic
- Thick = diastolic
- Ejection fraction
- Exercise/Stress
Testing
- Evaluate
for underlying coronary disease, as well as potential candidates for
transplantation
- Patients with a peak oxygen uptake (VO2) < 14 mL/kg/min have better outcomes with transplanted
- Evaluate
for underlying coronary disease, as well as potential candidates for
transplantation
- Coronary
angiography
- Not strongly recommended as part of the work-up, but can be useful to evaluate for underlying coronary disease and get an accurate ejection fraction
Classification and Grading



Current Nomenclature
Heart failure with reduced ejection fraction (HFrEF)
- Systolic
- < 40% EF
Heart failure with preserved ejection fraction (HFpEF)
- Diastolic
- > 50% EF
Management
- Heart failure with reduced ejection
fraction (HFrEF)
- Lifestyle Modifications
- Smoking cessation
- Restrict sodium to < 3g/day
- Restrict fluid to < 2L/day
- Pharmacotherapy
- Loop diuretic (if overload is
present)
- Furosemide, bumetanide, torsemide
- ACE inhibitors
- Lisinopril, enalapril
- Angiotension receptor-neprilysin
inhibitor (ARNI)
- NYHA II or III and:
- BNP > 150 ng/mL or hospitalized with last 12 months
- SBP > 100 mmHg
- GFR > 30 mL/min
- No history of angioedema
- NYHA II or III and:
- ARB
- Candesartan, valsartan
- Beta blockers
- Carvedilol, metoprolol, bisoprolol
- Mineralcorticoid receptor antagonist
(MRA)
- Spironolactone, eplerenone
- Selective sinus node inhibitor
- Ivabradine
- Need a resting HR > 70 bpm on maximum BB therapy
- Loop diuretic (if overload is
present)
- Ischemic heart disease
- Increase coronary perfusion
- Decrease myocardial demand
- Hyperlipidemia
- High-intensity statin
- Cardiac rehabilitation
- Lifestyle Modifications
- Heart failure with preserved
ejection fraction (HFpEF)
- Differences with HFrEF
- ACEI/ARB not as useful
- MRA used more often
- Diuretics OK but be careful for volume depletion
- Don’t use BB unless compelling indication
- Differences with HFrEF
References
- Tan LB, Williams SG, Tan DK, Cohen-Solal A. So many definitions of heart failure: are they all universally valid? A critical appraisal. Expert review of cardiovascular therapy. 2010; 8(2):217-28. [pubmed]
- CDC Heart Failure Data Sheet. https://www.cdc.gov/dhdsp/data_statistics/fact_sheets/fs_heart_failure.htm
- Emory Healthcare Heart Failure Statistics. https://www.emoryhealthcare.org/heart-vascular/wellness/heart-failure-statistics.html
- Mann DL, Chakinala M. Heart Failure: Pathophysiology and Diagnosis. In: Jameson J, Fauci AS, Kasper DL, Hauser SL, Longo DL, Loscalzo J. eds. Harrison’s Principles of Internal Medicine, 20e New York, NY: McGraw-Hill; . http://accessmedicine.mhmedical.com.ezproxy.uthsc.edu/content.aspx?bookid=2129§ionid=192028958
- Davie AP, Francis CM, Caruana L, Sutherland GR, McMurray JJ. Assessing diagnosis in heart failure: which features are any use? QJM : monthly journal of the Association of Physicians. 1997; 90(5):335-9. [pubmed]
- Kelder JC, Cramer MJ, van Wijngaarden J, et al. The diagnostic value of physical examination and additional testing in primary care patients with suspected heart failure. Circulation. 2011; 124(25):2865-73. [pubmed]
- Davie AP, Francis CM, Love MP, et al. Value of the electrocardiogram in identifying heart failure due to left ventricular systolic dysfunction. BMJ (Clinical research ed.). 1996; 312(7025):222. [pubmed]
- Maisel A. B-type natriuretic peptide levels: diagnostic and prognostic in congestive heart failure: what’s next? Circulation. 2002; 105(20):2328-31. [pubmed]
- Knudsen CW, Omland T, Clopton P, et al. Diagnostic value of B-Type natriuretic peptide and chest radiographic findings in patients with acute dyspnea. The American journal of medicine. 2004; 116(6):363-8. [pubmed]
- Badgett RG, Mulrow CD, Otto PM, Ramírez G. How well can the chest radiograph diagnose left ventricular dysfunction? Journal of general internal medicine. 1996; 11(10):625-34. [pubmed]
- Bart BA, Shaw LK, McCants CB, et al. Clinical determinants of mortality in patients with angiographically diagnosed ischemic or nonischemic cardiomyopathy. Journal of the American College of Cardiology. 1997; 30(4):1002-8. [pubmed]
- Ho KK, Pinsky JL, Kannel WB, Levy D. The epidemiology of heart failure: the Framingham Study. Journal of the American College of Cardiology. 1993; 22(4 Suppl A):6A-13A. [pubmed]
- Yancy CW, Jessup M, Bozkurt B, et al. 2013 ACCF/AHA guideline for the management of heart failure: executive summary: a report of the American College of Cardiology Foundation/American Heart Association Task Force on practice guidelines. Circulation. 2013; 128(16):1810-52. [pubmed]
- Yancy CW, Jessup M, et al. 2016 ACC/AHA/HFSA Focused Update on New Pharmacological Therapy for Heart Failure: An Update of the 2013 ACCF/AHA Guideline for the Management of Heart Failure: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Failure Society of America. Circulation. 2016; 134(13):e282-93. [pubmed]
- Ponikowski P, Voors AA, Anker SD, et al. 2016 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: The Task Force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC)Developed with the special contribution of the Heart Failure Association (HFA) of the ESC. European heart journal. 2016; 37(27):2129-2200. [pubmed]
- Parch J, Powell C. No longer failing to treat heart failure: A guideline update review. JAAPA : official journal of the American Academy of Physician Assistants. 2019; 32(1):11-15. [pubmed]