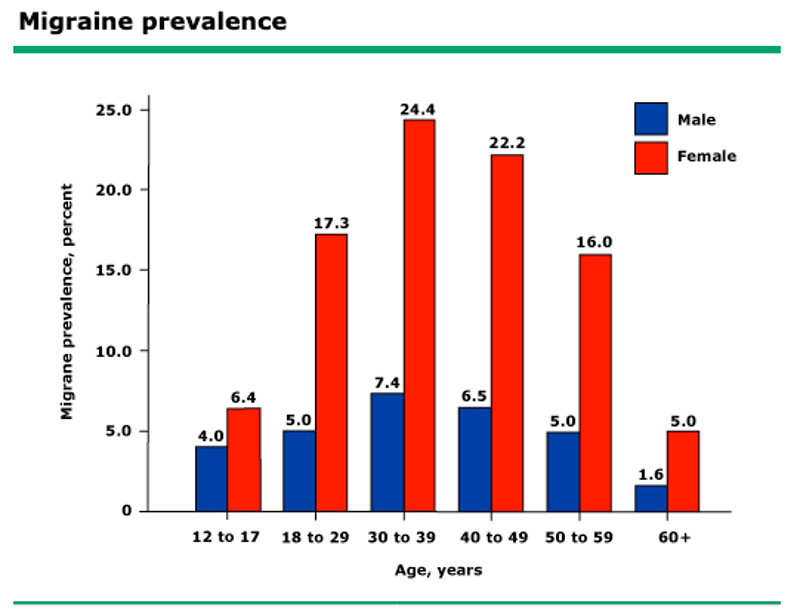
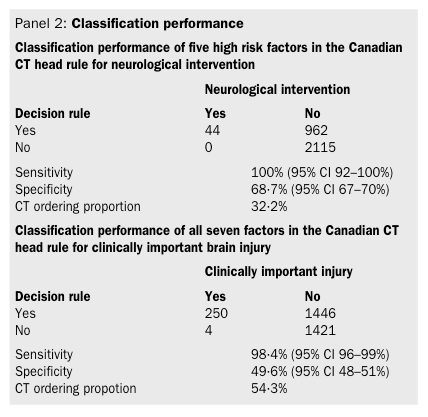
Wernicke’s Aphasia
Other Known Aliases – receptive aphasia
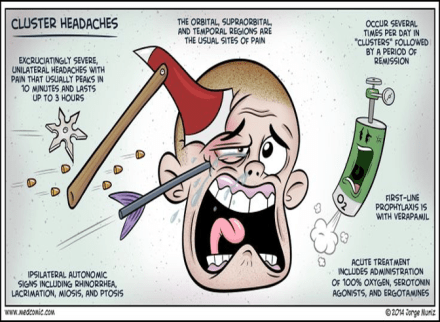
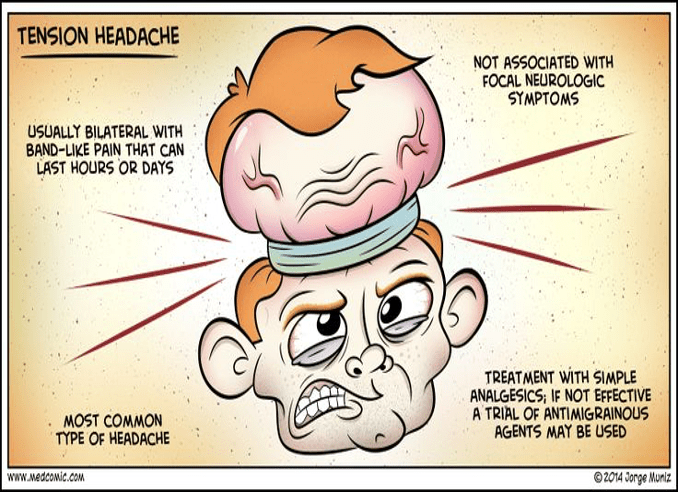
Definition – difficulty in understanding written or spoken language, but demonstrate fluent speech that lacks meaning
Clinical Significance – this condition manifests due to damage to Wernicke’s area of the brain in Brodmann area 22. This region is located in the posterior section of the superior temporal gyrus of the dominant hemisphere.

History – Named after Karl Wernicke (1848-1905), who was a German physician, anatomist, and neuropathologist and received his medical doctorate from the University of Breslau in 1870. He went on to study under Ostrid Foerster and Theodor Maynert after serving as an army surgeon during the Franco-Prussian War and had a modest career in both private and academic practice, culminating as head of the University Hospital’s Department of Neurology and Psychiatry at Breslau. A proponent of the same cerebral localization theory as Broca, he described his theory of “sensory aphasia” being different from Broca’s “motor phasia” in his book Der Aphasische Symptomencomplex in 1874.
References
- Firkin BG and Whitwirth JA. Dictionary of Medical Eponyms. 2nd ed. New York, NY; Parthenon Publishing Group. 1996.
- Bartolucci S, Forbis P. Stedman’s Medical Eponyms. 2nd ed. Baltimore, MD; LWW. 2005.
- Yee AJ, Pfiffner P. (2012). Medical Eponyms (Version 1.4.2) [Mobile Application Software]. Retrieved http://itunes.apple.com.
- Whonamedit – dictionary of medical eponyms. http://www.whonamedit.com
- Up To Date. www.uptodate.com
- Wernicke C. Der Aphasische Symptomencomplex. 1874.