***LISTEN TO THE PODCAST HERE***
Epidemiology
- 300,000-600,000 cases per year in the United States
- It is estimated that up to 50% will have post-thrombotic syndrome
Why Are We So Scared?
- As many as 20% of patients with 1st onset PTE have no identifiable risk factors
- 10-30% 1-month mortality with up to 25% presenting as sudden death
- Fear of litigation is #1 reason clinicians work-up low risk PTE
Why Can’t We Test Everyone?
- Up to $16,000 per patient in total health care costs
- 6 times more deaths with testing and treatment
Signs and Symptoms
The majority of the classic signs and symptoms come from PIOPED II study and EMPEROR registry. These include:
- Dyspnea (73%)
- Chest Pain (64%)
- Tachypnea (57%)
- DVT findings or leg pain/swelling (47%)
- Tachycardia (26%)
- Dizziness (12%)
- Hemoptysis (10%)
The EMPEROR registry took it a step further and determined mean vital sign measurements of:
- Heart rate – 95 bpm
- Respiratory rate – 20 bpm
- Oxygen saturation – 95%
A recent trail in the NEJM called PESIT concluded as many as 1 in 6 patients with first time syncope has a PTE on inpatient work-up. This study has been largely panned by the EM community and you can read their take from the links below:
- PulmCCM
- Rory Spiegel of EmCrit Part I and Part II
- Seth Trueger
- Ryan Radecki
- Simon Carly of St. Emlyn’s
- Patrick Bafuma of EM In Focus
- NEJM Resident 360 Discussion
- Salim Rezaie of REBEL EM
- Lauren Westafer and Jeremy Faust of FOAMCast
- Rejected Letter to Editor from respected PTE researcher Jeffrey Kline
- Brown Emergency Medicine
Pre-Test Probability Scores
The most well know score is the Wells Criteria first published in 1998 and then revised and simplified in 2000 and 2001.
A second calculation is the Geneva Score first published in 2001 and revised and simplified in 2006 and 2008.
The Pulmonary Embolism Rule-Out Criteria was published in 2008 by Jeff Kline and is a second set of criteria to definitively rule-out PTE in patients ALREADY SCORE AS LOW RISK by Wells or Geneva.
What about just good ol’ clinical gestalt? An interesting study was performed in 2013 looking at the accuracy of Wells vs Geneva vs Gestalt and found:
- Clinical gestalt had a lower missed rate of PTE in low-risk patients
- Clinical gestalt had a high accuracy of diagnosing PTE in high-risk patients
The Work-Up of Suspected PTE
- Electrocardiogram is not senstitive nor specific for PTE but should be ordered on every patient with chest pain and/or shortness of breath to rule-out ACS
- The EMCMD talks about the 10 ECG findings of PTE in the best video I have every scene
- The EMCMD talks about the 10 ECG findings of PTE in the best video I have every scene
- D-Dimer
- High senstivity = good for rule-out
- Should only be used after pre-test probability due to the false positives and unnecessary work-ups
- ADJUST-PE Study
- Found D-Dimer go up with age and created an age adjusted D-Dimer cutoff of:
- Age (yr) x 10 as diagnostic threshold
- Found D-Dimer go up with age and created an age adjusted D-Dimer cutoff of:
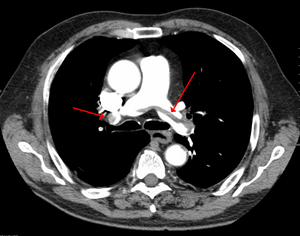
- Radiographic Imaging
- Computed tomography is gold standard but has higher radiation exposure and contrast loads
- Ventilation/Perfusion scan is safer in renal patients but up to 2/3rd are non-diagnostic

Risk Assessment
Once you diagnose a patient with a PTE, you have determine the patient’s risk and severity of disease.
- Echocardiogram
- Looking for RV strain
- RV:LV ≥ 1
- RV hypokinesis
- Paradoxical septal movement
- Tricuspid regurgitation
- Looking for RV strain
- Biomarkers
- Brain Natriuretic Peptide (BNP)
- > 90 pg/mL has been associated with increased mortality
- Troponin
- > 0.01 ng/mL suggests evidence of RV dysfunction
- Brain Natriuretic Peptide (BNP)
- Pulmonary Embolism Severity Index (PESI)
- Published in 2005 and simplified 2010
- Developed to help prognosticate 30d mortality and found low-risk patients (PESI – 0) can be safely treated as outpatient
Definitions/Grades of PTE
Treatment Strategies for PTE
- Anticoagulation
- Started with confirmation of PTE or with high pre-test probability during workup
- Lots of options (heparin, LMWH, direct thrombin inhibitors, Factor Xa inhibitors)
- Fibrinolytics
- Lots of recent research on who to lyse and who not to
- Original research showed benefit if full dose lytics were given to massive PTE, but harm in submassive patients
- This led to MOPPET in 2013 evaluating 1/2 dose lytics in submassive patients and found:
- Reduction in overall mortality
- No difference in bleeding complications
- Reduction in hospital stay
- PEITHO came next in 2014 and looked at full dose lytics vs anticoagulation only for submassive PTE and found:
- No mortality benefit
- Reduction in hemodynamic compromise
- Increase in major bleeding and intracranial hemorrhage

- Catheter Directed Therapy
- Good options in patients with a high risk of bleeding with systemic fibrinolytic therapy
- This can include:
- Focal fibrinolytic therapy at the clot
- Mechanical thombectomy
- Ultrasound Assisted Local Fibrinolytic Therapy
- ULTIMA and SEATTLE-II studies found reduction in RV:LV ratio and decreased bleeding complications
- Surgical Thrombectomy
- Can be used as a last resort option and mortality from these procedures has dramatically improved from 57% in the 1960s to < 6% in 2005
Putting It All Together
This is a graphic I modified from Jeff Kline and EmCrit that encompasses everything into a nice, neat package and as I have said before “algorithms will set you free”
References
- Beckman MG. Venous Thromboembolism: A Public Health Concern. Am J Prev Med. 2010;38(4S):S495-S501
- Spyropoulous AC. Direct medical costs of venous thromboembolism and subsequent hospital readmission rates: an administrative claims analysis from 30 managed care organizations. J Manag Care Pharm. 2007;13(6):475-486
- Calder KK. The mortality of untreated pulmonary embolism in emergency department patients. Ann Emerg Med. 2005;45(3):302-310
- Stein PD. Silent pulmonary embolism in patients with deep venous thrombosis: A systematic review. Am J Med. 2010;123:426-431
- Stein PD. Clinical Characteristics of patients with acute pulmonary embolism. Am J Med. 2007;120:871-879
- Pollack CV. Clinical characteristics, management, and outcoms of patients diagnosed with acute pulmonary embolism in the emergency department. JACC. 2011;57(6):700-706
- Wells PS. Use of a clinical model for safe management of patients with suspected pulmonary embolism. Ann Intern Med. 1998;129:997-1005
- Wells PS. Derivation of a simple clinical model to categorize patients probability of pulmonary embolism: increasing the models utility with the SimpliRED D-dimer. Thromb Haemost. 2000;83:416-420
- Wells PS. Excluding pulmonary embolism at the bedside without diagbnostic imaging: management of patients with suspected pulmonary embolism presenting to the emergency department by using a simple clinical model and D-dimer. Ann Intern Med. 2001;135:98-107
- van Belle A. Effectiveness of managing suspected pulmonary embolism using an algorithm combining clinical probability, D-dimer testing, and computed tomography. JAMA. 2006;295(2):172-179
- Wicki J. Assessing clinical probability of pulmonary embolism in the emergency ward: a simple score. Arch Intern Med. 2001;161:997-92-97
- Le Gal G. Prediction of pulmonary embolism in the emergency department: the revised Geneve score. Ann Intern Med. 2006;144:165-171
- Klok FA. Simplication fo the revised Geneva score for assessing clinical probability of pulmonary embolism. Ann Intern Med. 2008;168(19):2131-2136
- Kline JA. Prospective multicenter evaluation of the pulmonary embolism rule-out criteria. J Thromb Haemost. 2008;6:772-780
- Penaloza A. Comparison of the unstructured clinical gestalt, the wells score, and the revised Geneva score to estimate pretest probability for suspected pulmonary embolism. Ann Emerg Med. 2013;62(2):117-124
- Righini M. Age-adjusted D-dimer cutoff levels to rule-out pulmonary embolism: the ADJUST-PE study. 2014;311(11):1117-1124
- Stein PD. Clinical characteristics of patients with acute pulmonary embolism: data from IOPED II. Am J Med. 2007;120:871-879
- Anderson DR. Computerized tomographic pulmonary angiography versus ventilation perfusion lung scanning for the diagnosis of pulmonary embolism. Curr Opin Pulm Med. 2009;15:425–429
- Rudoni RR. Use of two-dimensional echocardiography for the diagnosi of pulmonary embolus. J Emerg Med. 1998;16(1):5-8
- Taylor RA. Point-of-care focused cardiac ultrasound for prediction of pulmonary embolism adverse outcomes. J Emerg Med. 2013;45(3):392-399
- Kiely DG. Elevated levels of natriuretic peptides in patients with pulmonary thromboembolism. Resp Med. 2005;99:1286-1291
- Jaff MR. Management of massive and submassive pulmonary embolism, iliofemoral deep vein thrombosis, chronic thromboembolic pulmonary hypertension: a scientific statement form the American Heart Association. Circulation. 2011;123:1788-1830
- Keller K. Cardiac troponin I for predicting right ventricular dysfunction and intermediate risk in patients with normotensive pulmonary embolism. Neth Heart J. 2015;23:55-61
- Aujesky D. Derivation and validation of a prognostic model for pulmonary embolism. Am J Respir Crit Care Med. 2005;172:1041-1046
- Jimenez D. Simplification of the pulmonary embolism severity index for prognostication in patients with acute symptomatic pulmonary embolism. Arch Intern Med. 2010;170(15):1383-1389
- Tapson VF. Treatment of pulmonary embolism: anticoagnulation, thrmbolytic therapy, and complications of therapy. Crit Care Clin. 2011;27:825-839
- Sharifi M. Moderate pulmonary embolism treated with thrombolysis. Am J Cardiol. 2013;111:273-277
- Zhang Z. Lower dosage of recombinant tissue-type plasminogen activator (rt-PA) in the treatment of acute pulmonary embolism: a systematic review and meta-analysis. Thrombosis Research. 2014;133:357-363
- Meyer GM. Fibrinolysis for patients with intermediate-risk pulmonary embolism. NEJM. 2014;370(15):1402-1411
- Chatterjee S. Thrombolysis for pulmonary embolism and risk of all-cause mortality, major bleeding, and intracranial hemorrhage: a meta-analysis. JAMA. 2014;311(23):2414-2421
- Curtis GM. Risk factors associated with bleeding after alteplase administration for pulmonary embolism: a case control study. 2014;34(8):818–825
- Kennedy RJ. Thrombus resolution and hemodynamic recovery using ultrasound-accelerated thrombolysis in acute pulmonary embolism. J Vasc Interv Radiol. 2013;24:841-848
- Kucher N. Randomized, controlled trial of ultrasound-assisted catheter-directed thrombolysis for acute intermediate-risk pulmonary pulmonary embolism. Circulation. 2014;129:479-486
- Cross FS. A survey of the current status of pulmonary embolectomy for massive pulmonary embolism. Circulation. 1967;35:186-191
- Stulz P. Decision making in the surgical treatment of massive pulmonary embolism. Eur J Cardio-thorac Surg. 1994;8:188-193
- Leacche M. Modern surgical treatment of massive pulmonary embolism: result in 47 sonsecutive patients after rapid diagnosis and aggressive surgical approach. J Thorac Cardiovasc Surg. 2005;129:1018-1023
- Prandoni P, et al (PESIT Investigators). Prevalance of Pulmonary Embolism amount Patients Hospitalized for Syncope. NEJM. 2016;375:1524-1531
- John MA, Klok FA, van Es N. D-dimer Interval Likelihood Ratios for Pulmonary Embolism. Acad Emerg Med. 2017;4;1-5.
- Konstantinides SV, et al. Impact of Thrombolytic Therapy on the Long-Term Outcome of Intermediate-Risk Pulmonary Embolism. JACC. 2017;69(12):1536-1544
- Sharifi M, et al (PEAPETT Investigators). Pulseless electrical activity in pulmonary embolism treated with thrombolysis. Am J Emerg Med. 2016;34(10):1963-1967
- Piazza G, et al. (SEATTLE-II Investigators). A prospective, single arm, multicenter trial of ultrasound-facilitated , catheter-directed, low-dose fibrinolysis for acute massive and submassive pulmonary embolism. JACC. 2015;8(1):1382-1392