Pulmonary
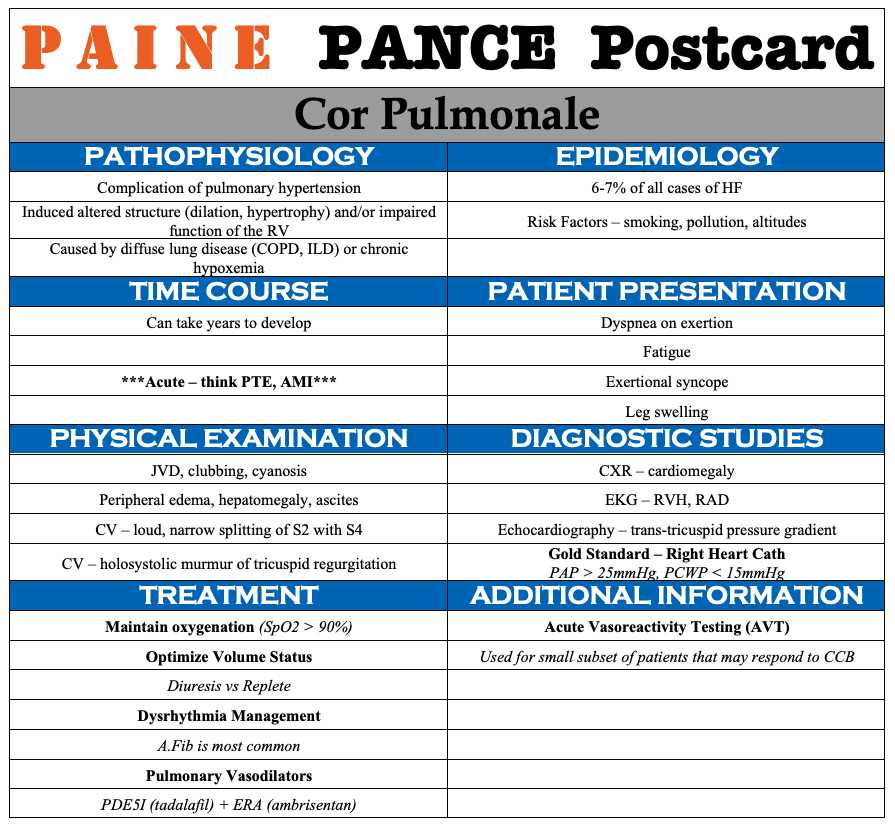
PAINE PANCE Postcard – Cor Pulmonale
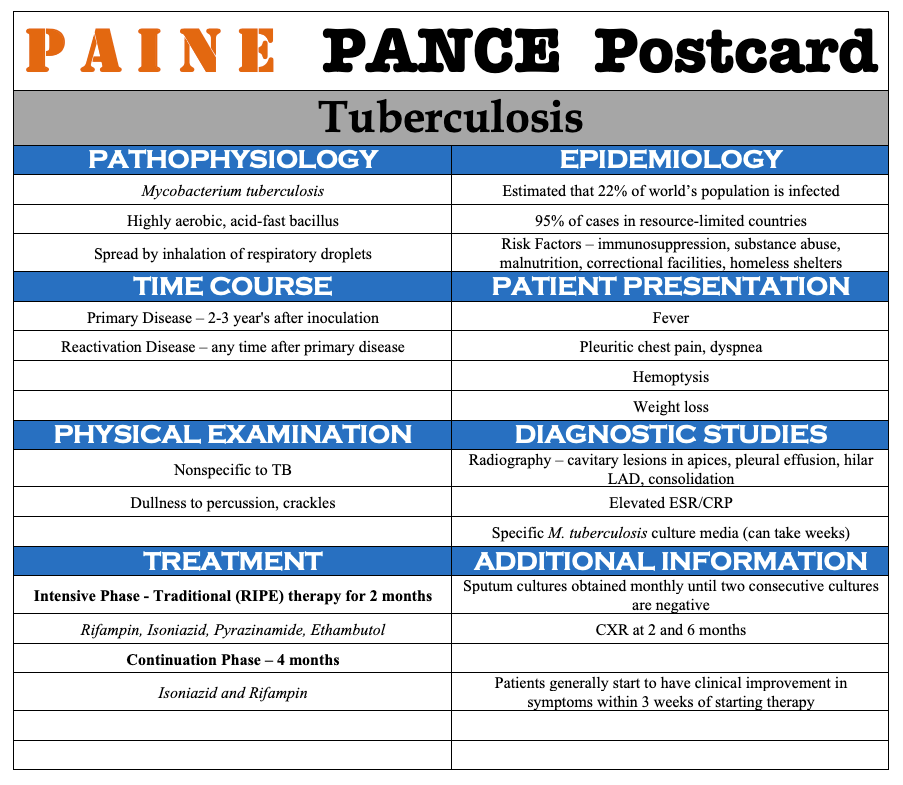
PAINE PANCE Postcard – Tuberculosis
PAINE PANCE Postcard – Croup
PAINE #PANCE Pearl – Pulmonology
Question
A 68yo man, with a history of COPD and 57-pack-year history of smoking, presents to the office with worsening shortness of breath on exertion, dizziness, and leg swelling over the past 6-months. He states that his inhalers don’t seem to be helping as much and he is finding it more and more difficult to walk without getting breathless. He denies orthopnea, paroxysmal nocturnal dyspnea, or increased cough. Vital signs show BP-157/98 mmHg, HR-88 bpm, RR-23 bpm, O2-90% on room air, and temp-98.9o. Physical examination reveals (+) JVD, widened split S2, 1+ pitting edema of the legs to the knee, and hepatomegaly. EKG is below.

- What does the EKG show?
- What is the diagnosis?
- What is the next step in evaluation?
- What is the definitive diagnostic study for this condition?
Answer
- The EKG shows numerous findings of chronic pulmonary disease including:
- Right axis deviation
- Peaked P-waves (>2.5mm) in inferior leads (II, III, aVF)
- Clockwise rotation of the heart with delayed R/S transition point
- Absent R-waves in right precordial leads (V1-V3)
- Low voltage left sided leads
- Given the absence of left sided heart failure and the patients extensive pulmonary history, the presentation is highly suggestive of pulmonary hypertension with cor pulmonale.
- Cor pulmonale is a complication of pulmonary hypertension and is defined as structural alterations or impaired function of the right ventricle
- The next step in evaluation of this patient is getting an echocardiogram to evaluate global heart function and degree of pulmonary hypertension present
- The definitive diagnostic study of pulmonary hypertension and cor pulmonale is a right heart catheterization.

References
Ep-PAINE-nym
Schamroth Test
Other Known Aliases – none
Definition – test used for diagnosing nail clubbing by examining the window seen between the nails of opposite fingers

Clinical Significance – there should be a diamond size window in “normal” nails when performing this test. In patients with nail clubbing, this window is obliterated
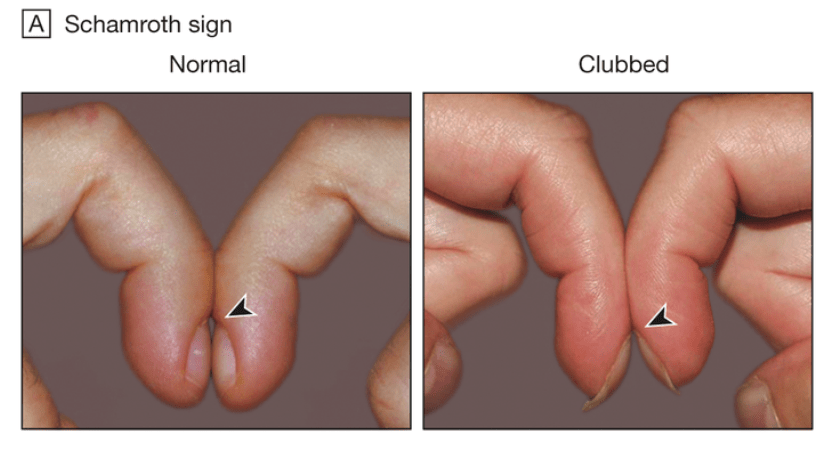
History – Named after Leo Schamroth (1924-1988), a Belgium-born, South African cardiologist who received his medical doctorate from the University of Witwatersrand in 1948. He completed his residency at Johannesburg General Hospital and joined the staff at Baragwanath Hospital in 1956, where he would stay his entire career culminating in chief physician in 1972. He would publish over 300 papers and eight textbooks in the realm of cardiology, and his Introduction to Electrocardiography is still one of the most stolen medical textbooks in the world. He published his eponymous finding in an article entitled “Personal Experience” in 1972 in the South African Medical Journal.

References
- Firkin BG and Whitwirth JA. Dictionary of Medical Eponyms. 2nd ed. New York, NY; Parthenon Publishing Group. 1996.
- Bartolucci S, Forbis P. Stedman’s Medical Eponyms. 2nd ed. Baltimore, MD; LWW. 2005.
- Yee AJ, Pfiffner P. (2012). Medical Eponyms (Version 1.4.2) [Mobile Application Software]. Retrieved http://itunes.apple.com.
- Whonamedit – dictionary of medical eponyms. http://www.whonamedit.com
- Up To Date. www.uptodate.com
- Schamroth L. Personal experience. S Afr Med J. 1976; 50(9):297-300. [pubmed]
PAINE #PANCE Pearl – Pulmonology
Question
A 68yo man, with a history of COPD and 57-pack-year history of smoking, presents to the office with worsening shortness of breath on exertion, dizziness, and leg swelling over the past 6-months. He states that his inhalers don’t seem to be helping as much and he is finding it more and more difficult to walk without getting breathless. He denies orthopnea, paroxysmal nocturnal dyspnea, or increased cough. Vital signs show BP-157/98 mmHg, HR-88 bpm, RR-23 bpm, O2-90% on room air, and temp-98.9o. Physical examination reveals (+) JVD, widened split S2, 1+ pitting edema of the legs to the knee, and hepatomegaly. EKG is below.

- What does the EKG show?
- What is the diagnosis?
- What is the next step in evaluation?
- What is the definitive diagnostic study for this condition?
Ep-PAINE-nym
Hippocratic Fingers
Other Known Aliases – clubbing of the fingers
Definition – soft tissue swelling of the terminal phalynx
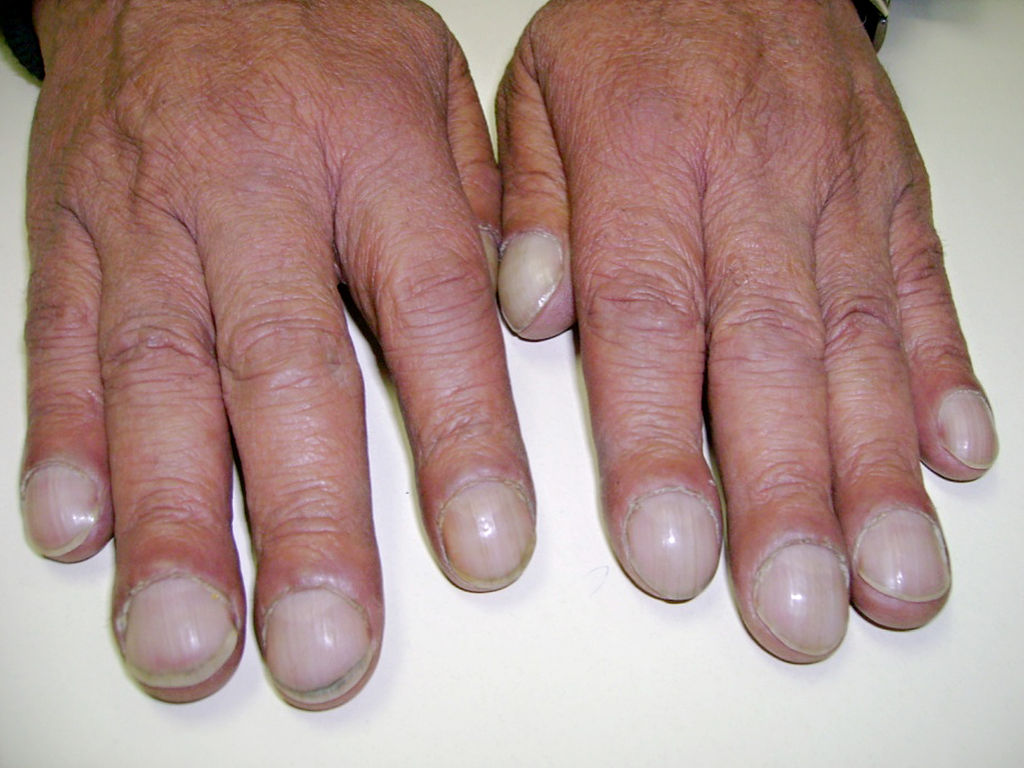
Clinical Significance – this is a nonspecific clinical finding that can be caused by numerous diseases, but is classically associated with any disease that can cause physiologic hypoxia. The exact mechanism for development is unknown, but is theorized to be caused by vasodilation, secretion of growth factors, overproduction of prostaglandin E2, and increased entry of megakaryocytes.
History – Named after Hippocrates (460-370 BC), who was a Greek physician and is considered to be the “Father of Medicine”, due to his approach in observation of clinical diseases. Though I cant find an original source for this eponym, it is one of those historical “facts” that nobody really challenges.

References
- Firkin BG and Whitwirth JA. Dictionary of Medical Eponyms. 2nd ed. New York, NY; Parthenon Publishing Group. 1996.
- Bartolucci S, Forbis P. Stedman’s Medical Eponyms. 2nd ed. Baltimore, MD; LWW. 2005.
- Yee AJ, Pfiffner P. (2012). Medical Eponyms (Version 1.4.2) [Mobile Application Software]. Retrieved http://itunes.apple.com.
- Whonamedit – dictionary of medical eponyms. http://www.whonamedit.com
- Up To Date. www.uptodate.com
Ep-PAINE-nym
Ghon Focus and Complex
Other Known Aliases – none
Definition – radiographic finding in primary tuberculosis where cellular and biochemical reaction to the infection forms a nodular granulomatous structure (focus) which can enlarge and invade adjacent lymphatics and hilar lymph nodes (complex).
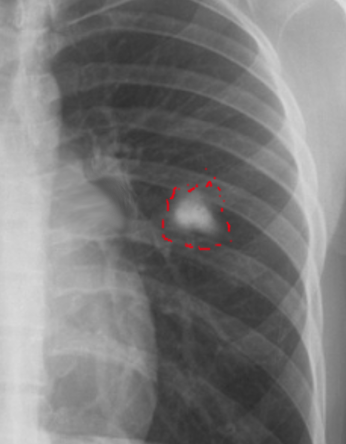
Clinical Significance – this finding on radiography is pathognomonic for primary active tuberculosis
History – Named after Anton Ghon (1866-1936), who was an Austrian pathologist and recieved his medical doctorate from the University of Graz in 1890. He would spend his entire career in pathology and bacteriology culminating in full professorship at the University of Prague in 1910. He frist published his eponymous findings in his 1912 work entitled “Der primäre Lungenherd bei der Tuberkulose der Kinder”. Unfortunately, we would go on to die from tuberculous pericarditis in 1928

References
- Firkin BG and Whitwirth JA. Dictionary of Medical Eponyms. 2nd ed. New York, NY; Parthenon Publishing Group. 1996.
- Bartolucci S, Forbis P. Stedman’s Medical Eponyms. 2nd ed. Baltimore, MD; LWW. 2005.
- Yee AJ, Pfiffner P. (2012). Medical Eponyms (Version 1.4.2) [Mobile Application Software]. Retrieved http://itunes.apple.com.
- Whonamedit – dictionary of medical eponyms. http://www.whonamedit.com
- Up To Date. www.uptodate.com
- Der primäre Lungenherd bei der Tuberkulose der Kinder. Berlin & Wien, Urbach & Schwarzenberg, 1912.
- Ober WB. Ghon but not forgotten: Anton Ghon and his complex. Pathol Annu. 1983; 18 Pt 2:79-85. [pubmed]
PAINE #PANCE Pearl – Pulmonology
Question
Carcinoid lung tumors are a rare pulmonary malignancy and have classic, characteristics signs and symptoms associated with them.
- What are the PULMONARY specific symptoms?
- What are the classic SYSTEMIC symptoms of carcinoid syndrome?

Answer
- Carcinoid lung tumors typically arise in the proximal airways and patienta can have obstructing symptoms such as dyspnea, cough, wheezing, chest pain, and recurrent pneumonia due to impaired sputum clearance. These tumors are also hypervascular and hemoptysis is also common.
- Since carcinoid tumors are neuroendocrine tumors they produce and secrete a host of vasoactive substances that can cause a host of specific signs and symptoms. The classic presentation for carcinoid syndrome include flushing, telangiectasias, diarrhea, and bronchospasm