PAINE PANCE Postcard – Croup
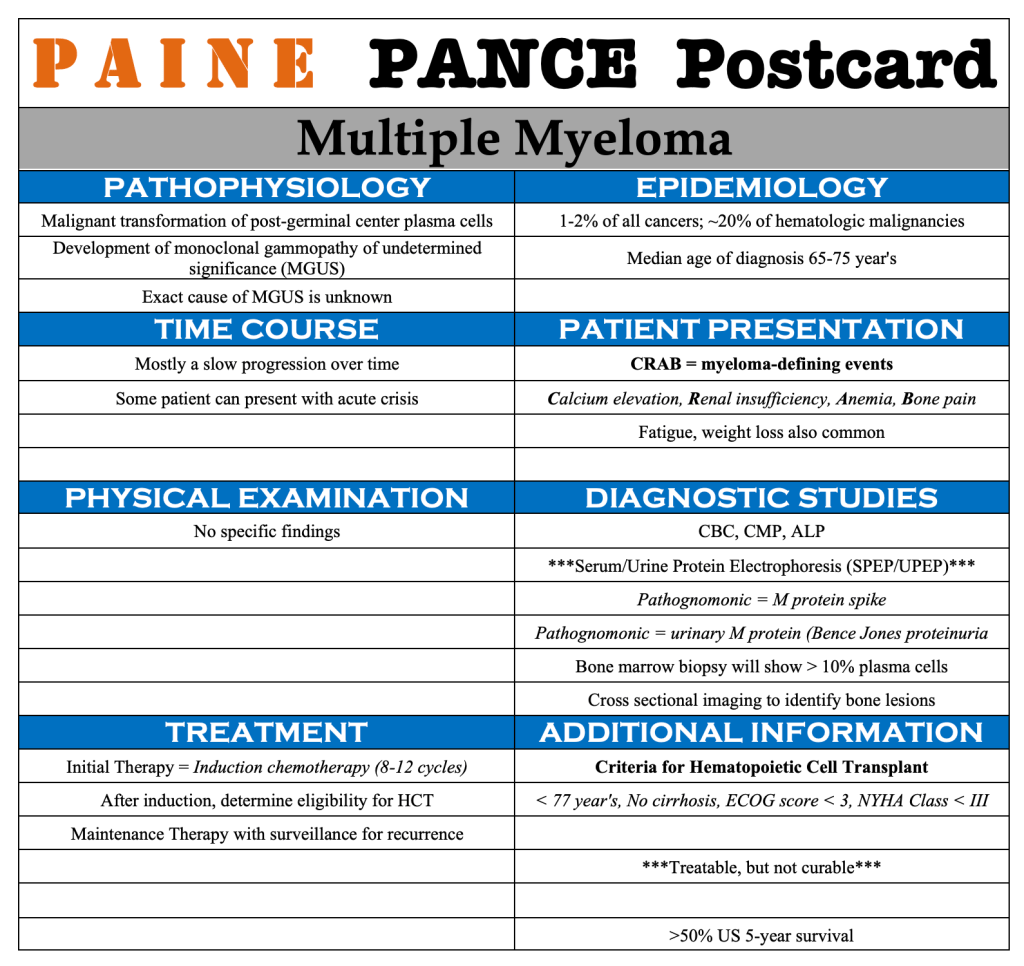
PAINE PANCE Postcard – Multiple Myeloma
PAINE PANCE Postcard – Hodgkin Lymphoma
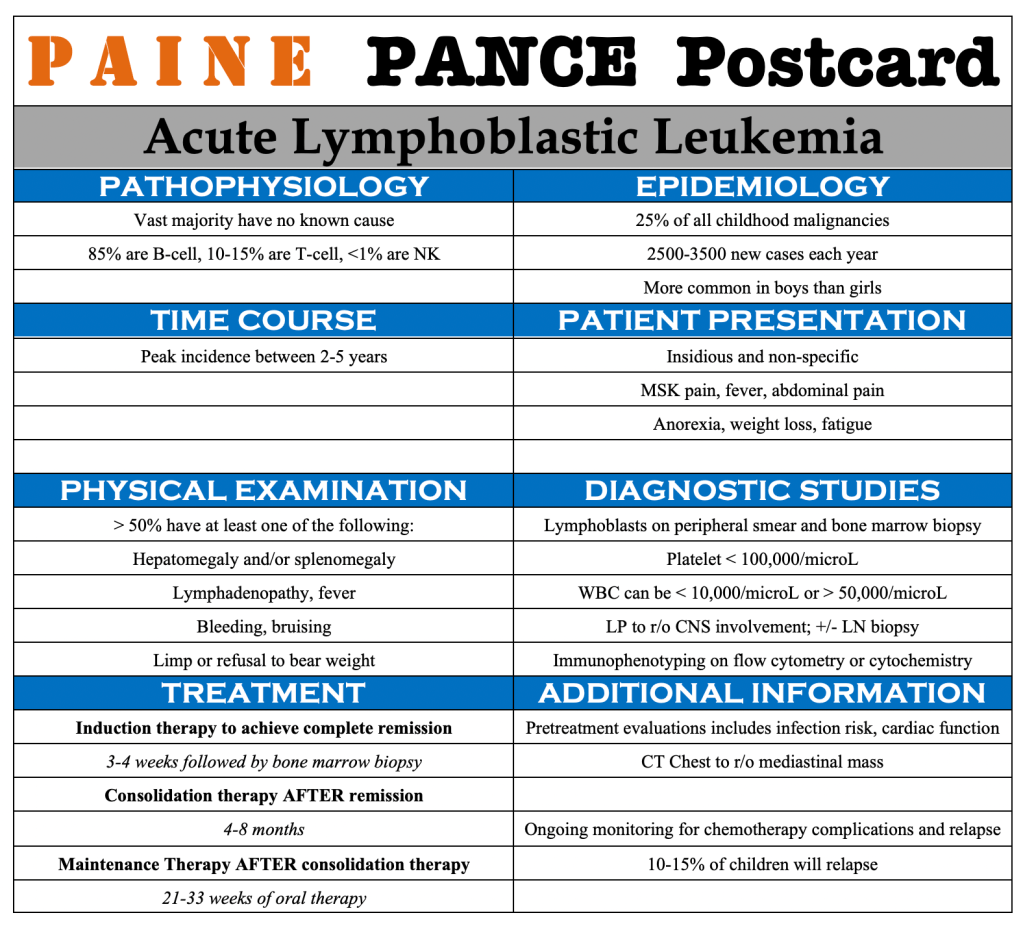
PAINE PANCE Postcard – Acute Lymphoblastic Leukemia

OBGYN – Placental Abruption

OBGYN – Placenta Previa

Cardiovascular – Cardiogenic Shock

Cardiovascular – AV Blocks